CH 301 Lecture Notes - Lecture 7: Principal Quantum Number, Rydberg Formula, Azimuthal Quantum Number
Document Summary
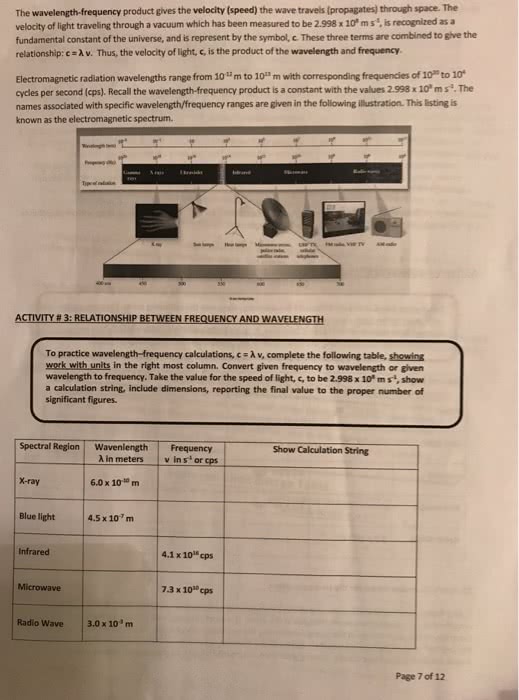
Wavelength is measure of the distance covered by the wave (nm or m) The frequency (v) is the number of waves that pass a point in a given period of time (1/s or hz) Periodic changes of electric and magnetic fields in time and space. Two fields are always perpendicular to each other. C is a constant in a vacuum (wavelength)(frequency)=c. Ideas about modern atomic theory stemmed from observations of the interactions between light and matter. Observations not explained well by classical mechanics or electromagnetism: discrete spectra of atoms, blackbody problem, photoelectric effect. Exciting gas atoms causes light to be emitted- bright light spectra . There is different emission spectra for different gases. Radiation emitted-light given out as electron returns to atom. Electromagnetic radiation emitted by an opaque or non-reflective body. Spectrum and intensity which depend on ly the temperature of the body. A method for determining the temperature of stars.