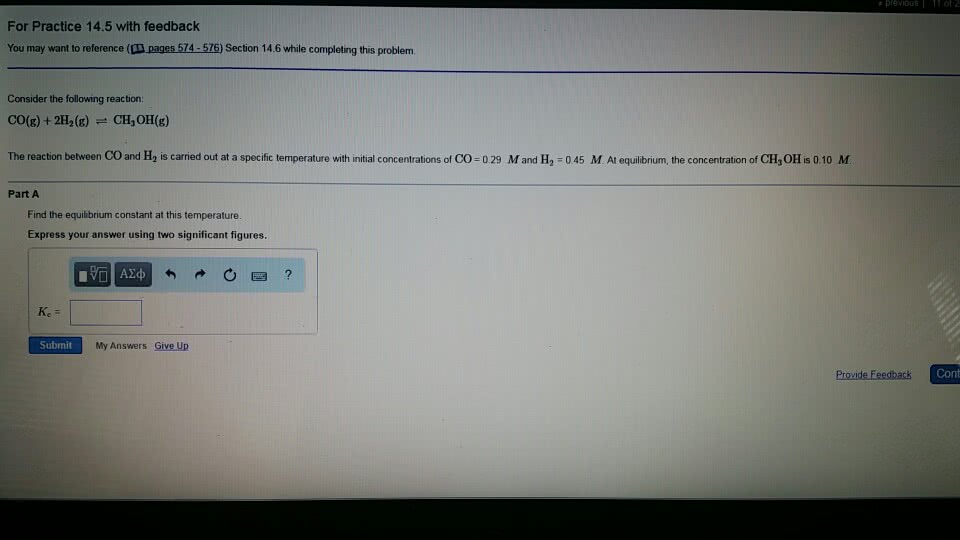
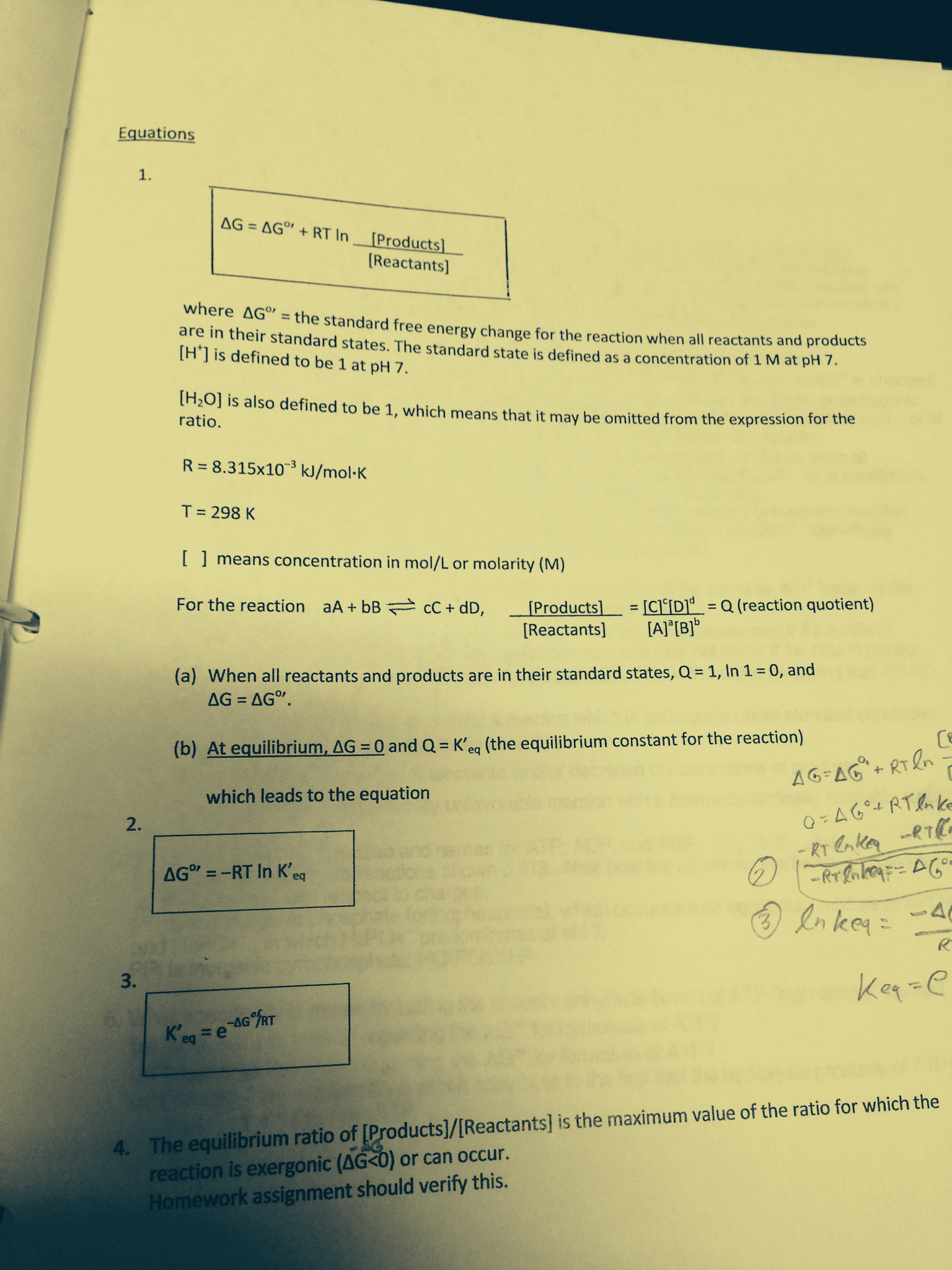1.) For a reaction system that is at equilibrium, which of the following must always be true?
a. q = 0 b. ?S = 0 c. ?G = 0 d. ?H = 0
2.) A certain reaction is found to be reactant favored. Which of the following is a correct description of the
reaction?
a. ?G° > 0, K > 1 b. ?G° > 0, K < 1 c. ?G° < 0, K > 1 d. ?G° < 0, K < 1
3.) What does the third law of thermodynamics state?
a. It states that a substance that is perfectly crystalline at 0 K has an entropy of zero.
b. It states that if two systems are each in thermal equilibrium with a third, they are also in thermal
equilibrium with each other.
c. It states that any quantity which has a continuous differentiable symmetry in the action has an
associated conservation law.
d. It states that the increase in internal energy of a closed system is equal to the difference between
the heat supplied to the system and the work done by the system.