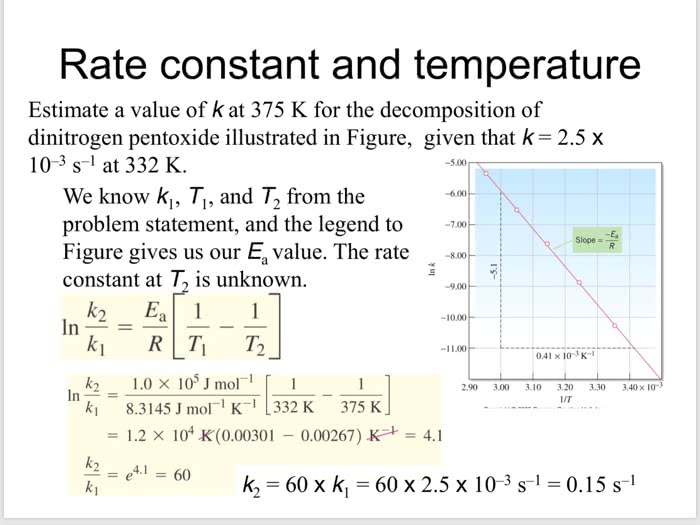
Now you try it Only...Please and Thank You
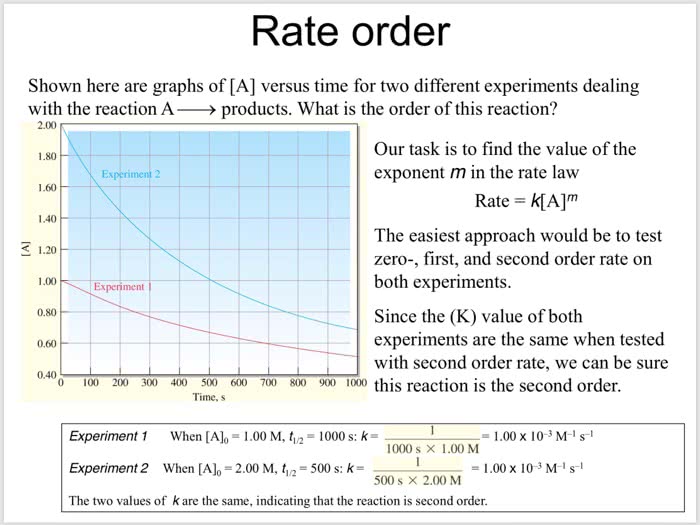
Rate order Shown here are graphs of [A] versus time for two different experiments dealing with the reaction A> products. What is the order of this reaction? 2.00 Our task is to find the value of the exponent m in the rate law 1.80 Experiment 2 1.60 Rate kA]m 1.40 The easiest approach would be to test zero-, first, and second order rate on both experiments S 1.20 1.00 ment 0.80 Since the (K) value of both experiments are the same when tested with second order rate, we can be sure 0.60 0.40 0 100 200 300 400 500 600 700 800 900 1000 this reaction is the second order Time, s = 1.00 x 10 3 M-IS-I Experimen When [Alo 1.00 M, t12 1000 s: k- Experiment 2 When [A],-2.00 M, t12-500 s: k= The two values of k are the same, indicating that the reaction is second order 1000 sà 1.00 M = 1.00 x 10-3 M-IS-I 500 s à 2.00 M