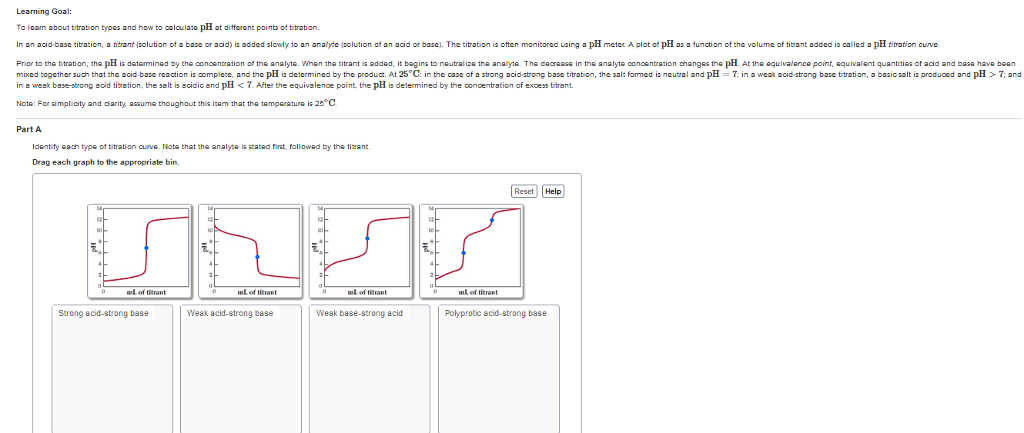
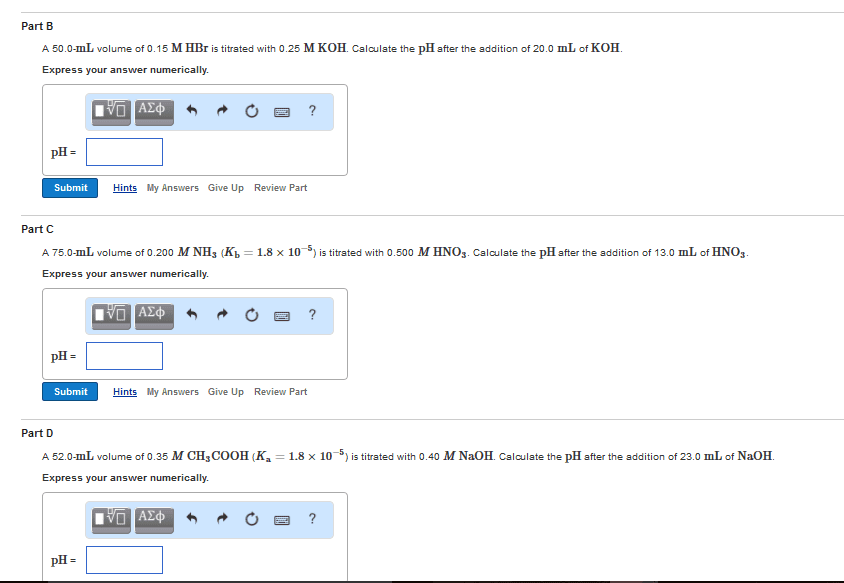
CH 302 Lecture Notes - Lecture 1: Houghton Mifflin Harcourt, Titration Curve, Bromothymol Blue
Document Summary
Acid/base titration is used to determine the concentration of an acid or a base in an unknown solution. The solution with the unknown concentration is called the analyte. The solution with the known concentration is called the titrant. To a specific volume of the analyte we use a buret to slowly add just enough titrant to react with all the analyte. This is called the stoichiometric point or the equivalence point. An indicator is used to help us detect the equivalence point of the titration. We can also use a ph meter to follow the ph changes and plot to produce a titration curve. From the plot, we determine the equivalence point. Equivalence point: the point at which chemically equivalent amounts (stoichiometric amounts) of reactants have reacted. End point: the point at which an indicator changes color and the titration should be stopped.