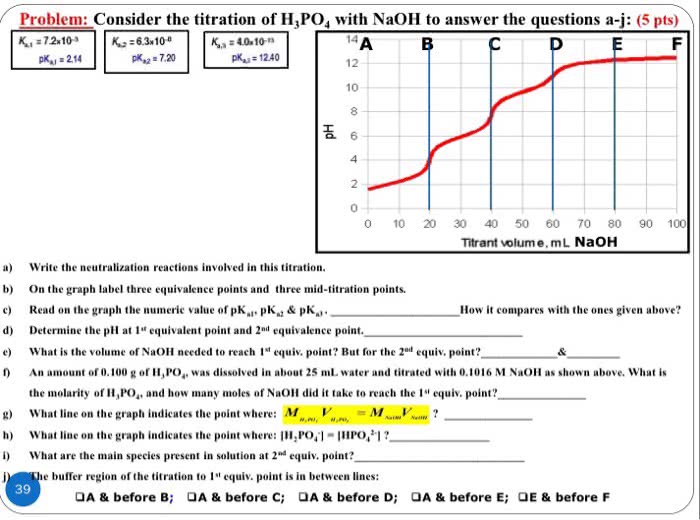
Problem: Consider the titration of H,PO4 with NaOH to answer the questions a-j: (5 pts) 14 pKu 12.40 pKAI 214 12 10 10 20 30 40 50 60 70 80 90 100 Titrant volume. mL NaOH a) Write the neutralization reactions involved in this titration. b) On the graph label three equivalence points and three mid-titration points. How it compares with the ones given above? c) Read on the graph the numeric value of pK pK & pKa. d) Determine the pH at l" equivalent point and 2"d equivalence point. e) What is the volume of NaoH needed to reach 1 equiv, point? But for the 2nd equiv. point & An amount of 0.100 g of HaPO,. was dissolved in about 25 mL water and titrated with 0.1016 M NaOH as shown above. What is the molarity of H,PO.. and how many moles of NaoH did it take to reach the 1" equiv. point? go What line on the graph indicates the point where: M MY h) What line on the graph indicates the point where: II,PO HPO I? What are the main species present in solution at 2"a equiv. point? jX The buffer region of the titration to 1" equiv. point is in between lines: 39 A & before B A & before C A & before D A & before E; OE & before F