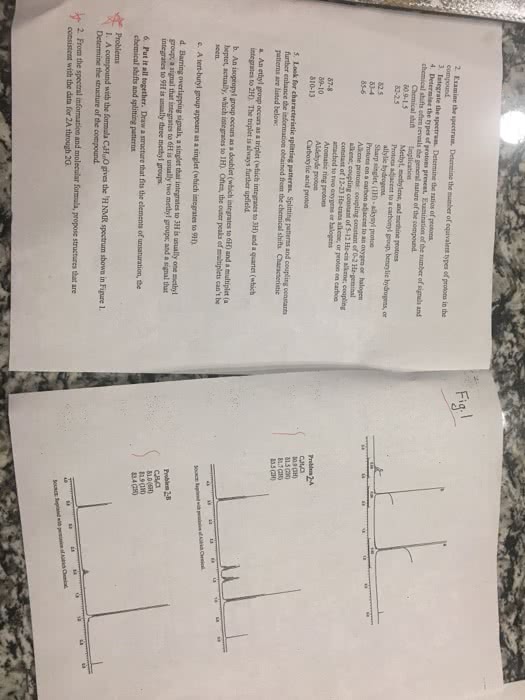
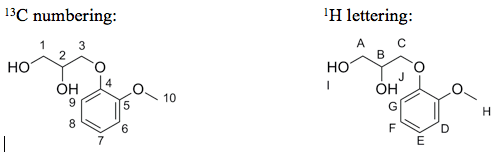The 1H and 13C NMR peaks of guaifenesin are: 1H NMR (CDCl3) δ 2.58 (t, 1H), 3.30 (d, 1H), 3.75â3.9 (m, 5H), 4.03â4.20 (m, 3H), 6.86â7.05 (m, 4H); 13C NMR (CDCl3) δ 56.1, 64.1, 70.4, 72.0, 112.1, 114.8, 121.4, 122.3, 148.2, 149.8. Using the labeled guaifenesin figures that follow, answer these questions:
a. In the 13C NMR of guaifenesin, what peaks (in ppm) correspond to carbons 4 and 5? What numbered carbons correspond to the peaks between 112.1â122.3 ppm?
b. Depending on the solvent used to take the 1H NMR, you can sometimes observe coupling to the protons attached to heteroatoms such as oxygen and nitrogen. In the 1H NMR of guaifenesin, what lettered hydrogen atoms correspond specifically to the 1H triplet at 2.58 ppm and the 1H doublet at 3.30 ppm?
c. In theory, the hydrogen atoms located at position H should show up as a singlet, but there are no singlets in the 1H NMR spectrum of guaifenesin. Why do we not observe a singlet for the hydrogen atoms at position H? Consider practical issues that may surface when running an NMR of a complex compound. 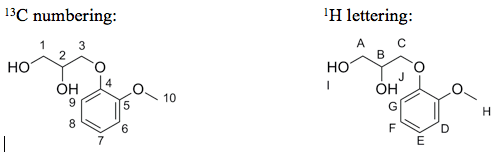
sC numbering: H lettering: 3 A C 2 HO OH4 OH â©10 8