Please help. Owl is picky about where the arrows go. Please be clear. Thank you!
Owl says there is an error with this one? Can you help?
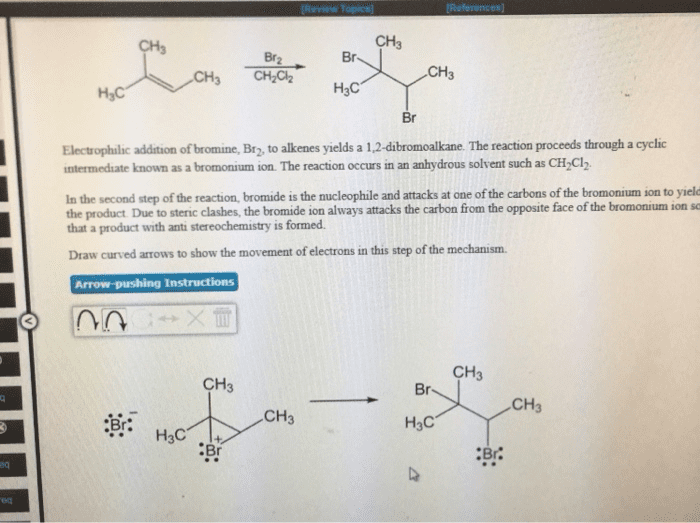
9% CH3 Br2 Br CH3 3 CH2C2 H3C Br Electrophlic addition of bromine, Br2, to alkenes yields a 1.2-dibromoalkane. The reaction proceeds through a cyclic intermediate known as a bromonium ion. The reaction occurs in an anhydrous solvent such as CH2Cl2 In the second step of the reaction, bromide is the nucleophile and attacks at one of the carbons of the bromonium ion to yield the product Due to steric clashes, the bromide ion always attacks the carbon from the opposite face of the bromonium ion se that a product with anti stereochemistry is formed Draw curved arrows to show the movement of electrons in this step of the mechanism. Instructions CH3 CH3 Br CH3 CH3 :Br. H3C .Br :Br. eq
Topics) Draw a stnactural formula for he maos product of he eaction shown Br2 H20 CH2 . Show product stereochemistry IF the reactant alkene has both carbons of the double bond within a ring Do not show stereochemistry in other cases. If the reaction produces a racemic mixture, just draw one stereoisomer
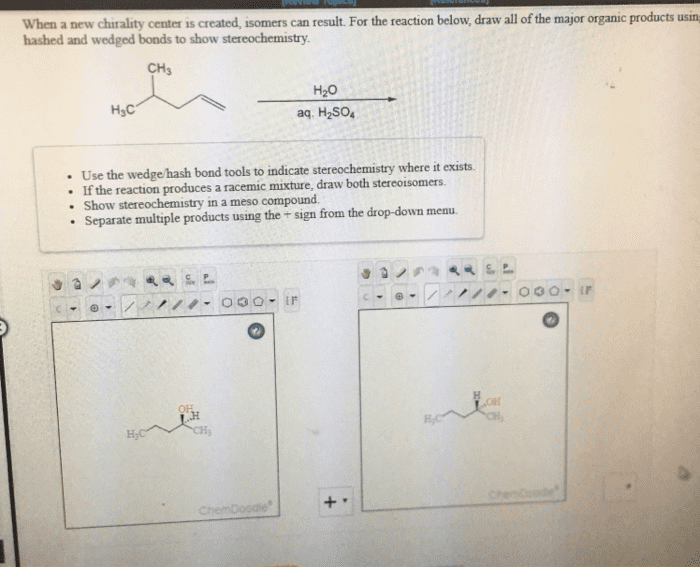
When a new chirality center is created, isomers can result. For the reaction below, draw all of the major organic products usin hashed and wedged bonds to show stereochemistry CH3 H20 aq. H2SO4 H3C Use the wedge/hash bond tools to indicate stereochemistry where it exists If the reaction produces a racemic mixture, draw both stereoisomers. e Show stereochemistry in a meso compound Separate multiple products using the + sign from the drop-down menu. C P c //. OOO. Ir 000. IF On
Show transcribed image text 9% CH3 Br2 Br CH3 3 CH2C2 H3C Br Electrophlic addition of bromine, Br2, to alkenes yields a 1.2-dibromoalkane. The reaction proceeds through a cyclic intermediate known as a bromonium ion. The reaction occurs in an anhydrous solvent such as CH2Cl2 In the second step of the reaction, bromide is the nucleophile and attacks at one of the carbons of the bromonium ion to yield the product Due to steric clashes, the bromide ion always attacks the carbon from the opposite face of the bromonium ion se that a product with anti stereochemistry is formed Draw curved arrows to show the movement of electrons in this step of the mechanism. Instructions CH3 CH3 Br CH3 CH3 :Br. H3C .Br :Br. eq
Topics) Draw a stnactural formula for he maos product of he eaction shown Br2 H20 CH2 . Show product stereochemistry IF the reactant alkene has both carbons of the double bond within a ring Do not show stereochemistry in other cases. If the reaction produces a racemic mixture, just draw one stereoisomer
When a new chirality center is created, isomers can result. For the reaction below, draw all of the major organic products usin hashed and wedged bonds to show stereochemistry CH3 H20 aq. H2SO4 H3C Use the wedge/hash bond tools to indicate stereochemistry where it exists If the reaction produces a racemic mixture, draw both stereoisomers. e Show stereochemistry in a meso compound Separate multiple products using the + sign from the drop-down menu. C P c //. OOO. Ir 000. IF On 



