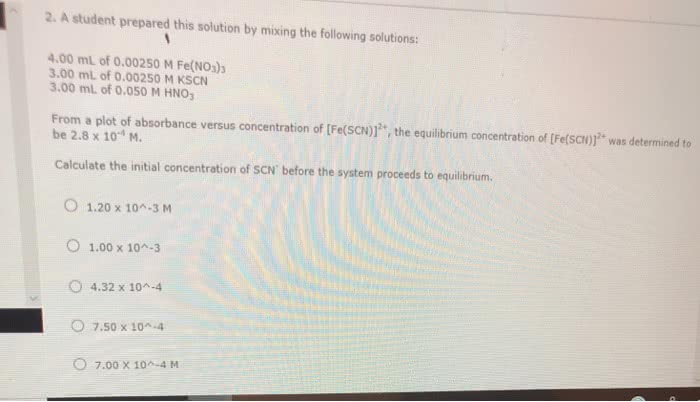
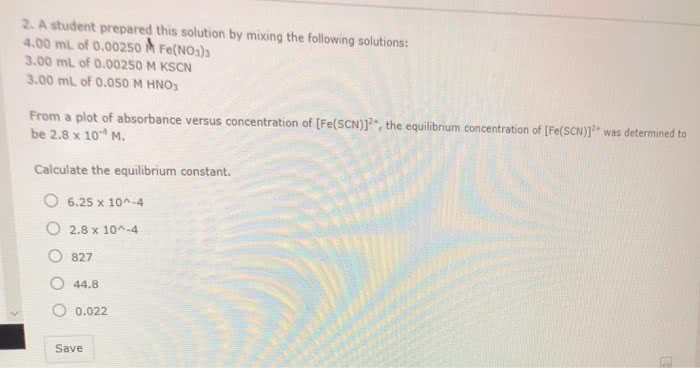
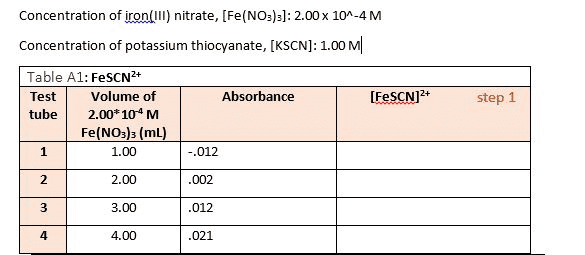
CHEM 103 Lecture Notes - Lecture 15: Iron(Iii) Nitrate, Potassium Thiocyanate, Limiting Reagent
Document Summary
Sample calculations : suppose that we prepare a mixture by mixing 2. 00 (cid:0) with 2. 00 (cid:0) Some red fescn2+ ion is formed by the complexation reaction. Its concentration at equilibrium is found to be 2. 50 (cid:0) 10 4 m. our problem is to find k for the reaction from this information. c. The initial concentrations of the reactants can be found from the following relation: initial concentration of reactants in mixture. For trial 1: initial concentration of fe3+ = 2. 00 (cid:0) initial concentration of scn = 2. 00 (cid:0) initial concentration of fescn2+ = 0. 00 m, because we did not add any to the mixture. Step 2: fill in the equilibrium concentration of product, fescn. [fescn2+]std = initial concentration of scn = 2. 00 (cid:0) Now you can calculate the equilibrium concentration of product in each of your four trials. Let"s use the fake data: abs of standard = 0. 045.