CHEM 110 Lecture Notes - Lecture 1: Special Relativity, Cathode Ray, Hubble Space Telescope
Document Summary
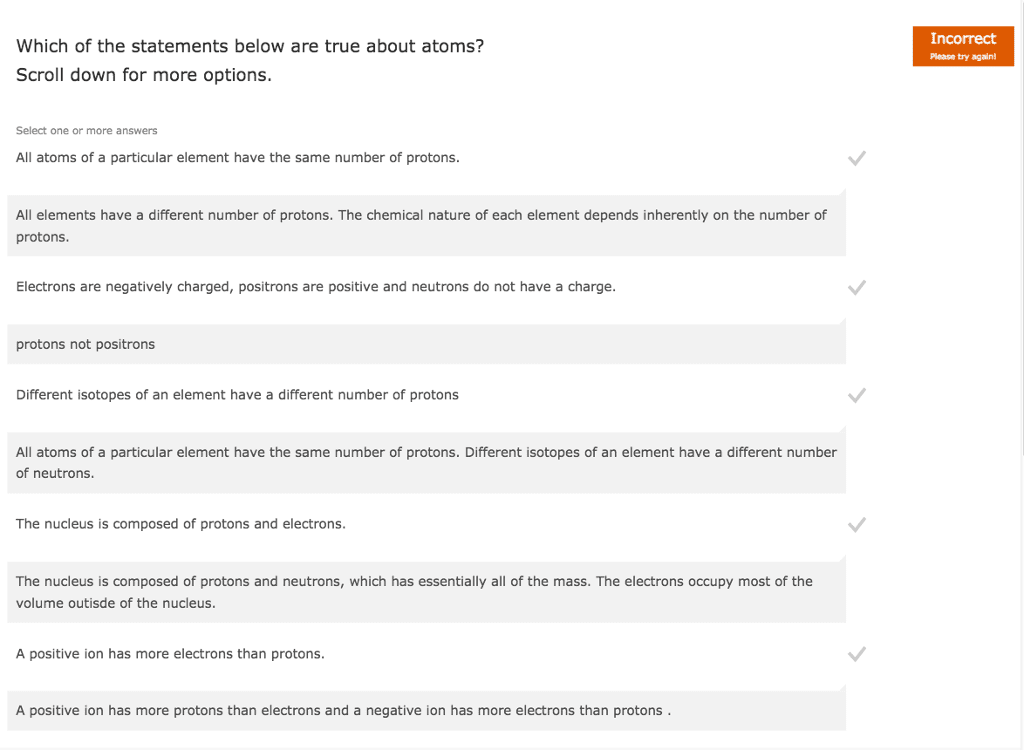
Chemistry is the study of matter and its changes. Atoms are the building blocks of all matter, and in the last week, we learned what an atom, supposedly the smallest particle, is actually made up of. An atom is composed of protons, neutrons, and electrons. Electrons have a negative one charge, but they also are elementary (or fundamental) particles. Protons and neutrons, however, are not elementary particles. There are up and down quarks, which are combined in the proton to create a positive one charge, and combined in a neutron to create a neutral, or zero, charge. Up quarks have a charge of positive 2/3, and down quarks have a charge of negative 1/3. A proton is made up of two up quarks and one down quark, equaling a positive one charge. A neutron is made up of two down quarks and one up quark, equaling a neutral, or zero, charge.