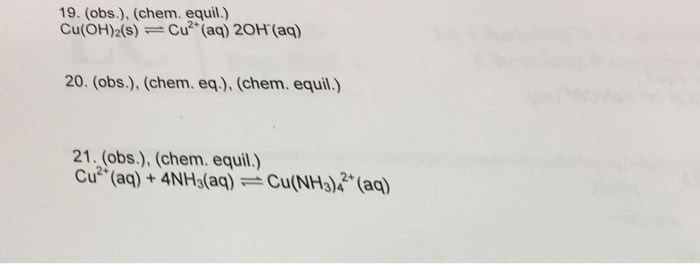University College - Chemistry Chem 112A Lecture Notes - Lecture 13: Sodium Chloride, Lead
16 views2 pages
15 Feb 2017
School
Department
Professor
Document Summary
0. 1m ki -> k+, i- in 1l of solution. Ksp = 1. 4 x 10-8 = s(0. 1 + 2s)2. I- is present in solution initially, which pushes the reaction left and there is less dissolution of. The presence of a common ion will result in a decrease of the solubility of the salt. But ag2cro4 is not soluble so first k2cro4 must be dissolved. Then agno3 is added creates ag2cro4 precipitate. Nacl added, so there is an excess of cl- ions. Original reaction pushed to the right, disrupting the equilibrium, and pulling the. Ag+ ions out of solution with cl- ions. The less soluble species is pulled out of solution, and forces the slightly non soluble species to dissolve. 0. 015m agno2 + 0. 005m pbno2 + nacl -> agcl + pbcl2. Agcl is insoluble (ksp = 1. 6 x 10-10) Pbcl2 is slightly soluble (ksp = 1. 6 x 10-5) If q = [pb2+][cl-]2 > ksp = 1. 6 x 10-5.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232