CHEM-UA 120 Lecture 1: Chemical formula, covalent bonds, molar mass
Document Summary
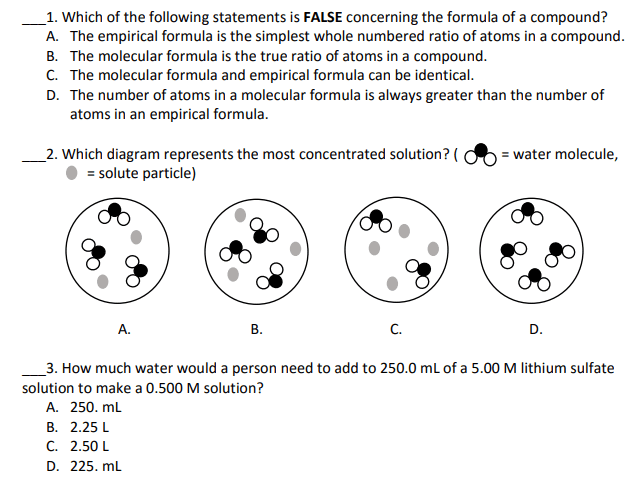
Are made from the symbols of their constituent elements. Are arranged in patterns that shows the ratio of atoms. Organic compounds are principally composed of carbon that is usually in combination with elements s, h, o and n (shon). Inorganic compounds all other compounds that are not indicated in organic compound. Like in math: examples: ho, ch2o, feso2, lewis electron formula aka electron-dot formula, shows the distribution of electrons in the outermost shell of the atom, electrons are represented by dots. Covalent bond - the second major type of atomic bonding occurs when atoms share electrons. Occurs because the atoms in the compound. Occurs because the toms in the compound have a similar tendency to gain electrons. Commonly occurs when two nonmetals bond together. Example of this is two hydrogen atoms and two fluorine atoms. *the other halogens (chlorine, bromine and iodine) and the common gases (nitrogen and oxygen)