CHEM 1307 Lecture Notes - Lecture 1: Lanthanide, Fluorine, Electronegativity
Document Summary
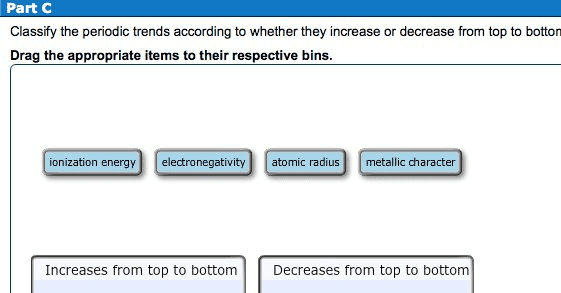
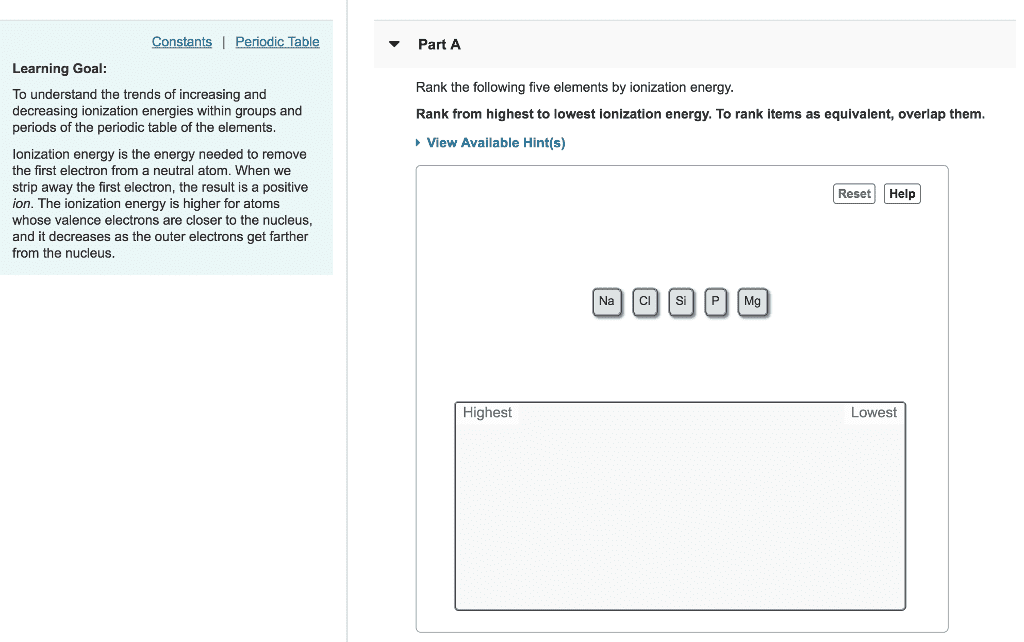
Theifelectrons btwtheoutermost electrons nucleus reduce force of attraction exerted on them by the nucleus. As you go from left toright in theperiodictable the outermost electrons are held more tightly because the no ofprotons keep increasing. The size of the atom atomicradius decreases t. From top to bottom in a periodic table the outermost electrons are held less tightly. From top to bottom in a periodic table to right in a periodic table electrons are added. The lanthanide contraction have higher ionization energies despite the fact that they have an additional shell of electrons. Ei gg tasedtwhen an electron is added to the valence shell of an atom. The addition of a first eletion to a neutral atom is usually exothermic. Itientigatiftistthe power of an atom in a molecule to attract electrons to itself. Electronegativity valves are obtained from thermochemical bond strengths. For elements in d block most electronegative 4 o least electronegative p blocks p blocks s.