CHEM 1030 Chapter 5: Thermochemistry

19
CHEM 1030 Full Course Notes
Verified Note
19 documents
Document Summary
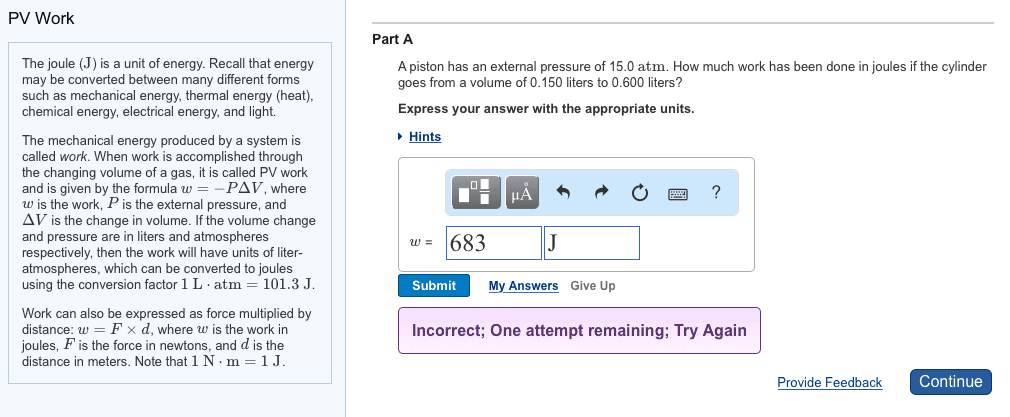
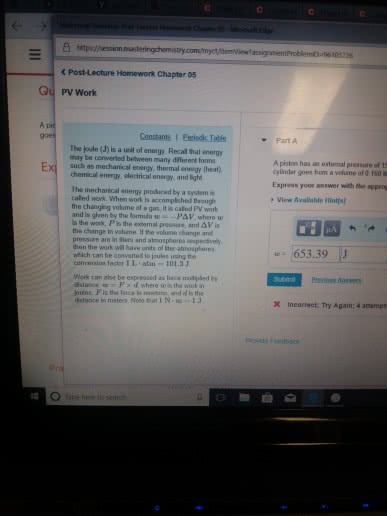
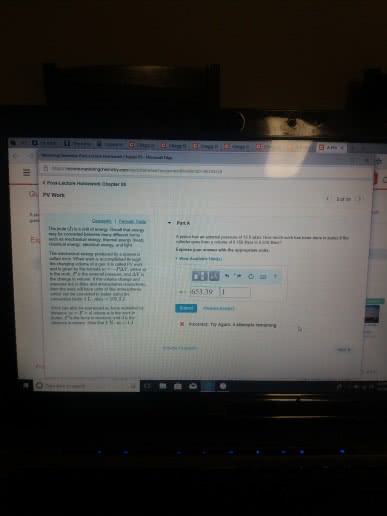
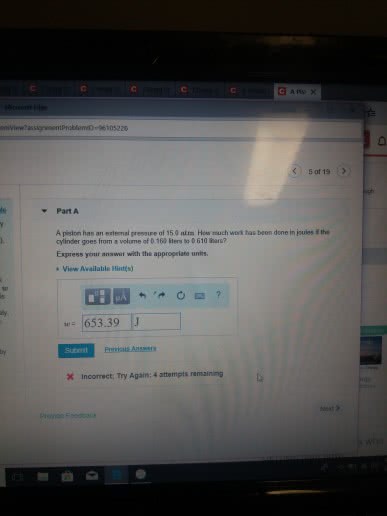
Potential energy - energy an object possesses by virtue of its position or chemical composition. Kinetic energy - energy an object possesses by virtue of its motion. Units of energy: the si unit of energy is the joule (j), an older, non-si unit is still in widespread use: the calorie (cal). System - includes the molecules we want to study. Work - energy used to move an object over some distance: w = f d, where w is work, f is the force, and d is the distance over which the force is exerted. Heat: energy can also be transferred as heat, heat flows from warmer objects to cooler objects. Conversion of energy - energy can be converted from one type to another. First law of thermodynamics: energy is neither created nor destroyed. In other words, the total energy of the universe is a constant; if the system loses energy, it must be gained by the surroundings, and vice versa.