CHEM123 Lecture Notes - Royal Aircraft Factory F.E.2, Electrochemistry, Galvanic Cell
63 views4 pages
30 Apr 2014
School
Department
Course
Professor
Document Summary
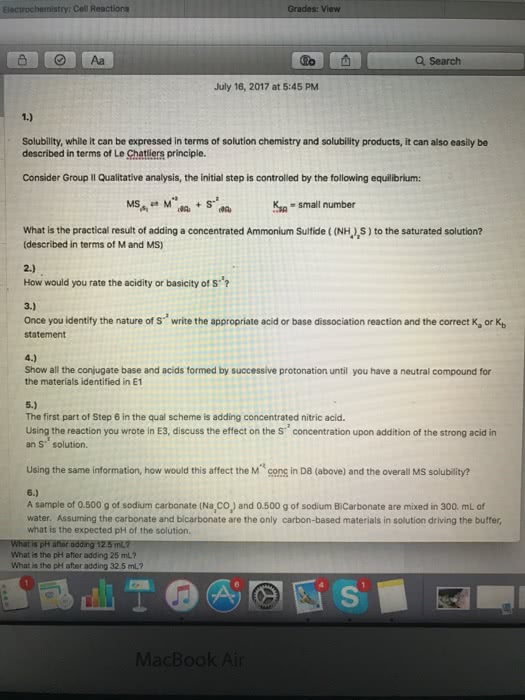
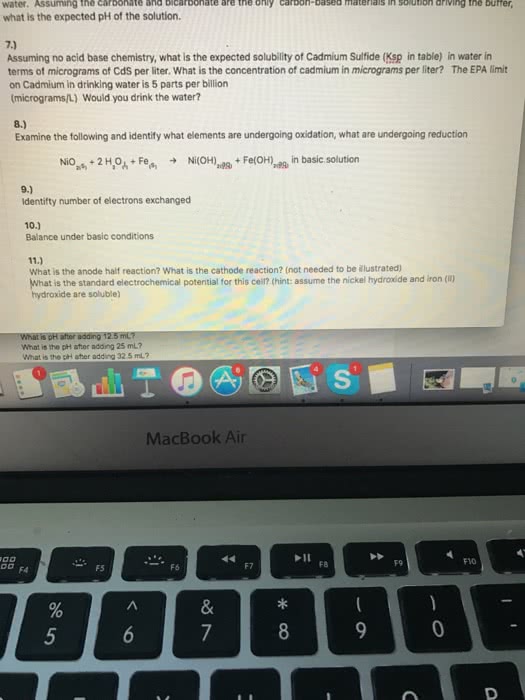
The equilibrium constants for the following reactions are given at 297 k. What is the correct shorthand notation for the cell depicted in the figure given below: pb(s) pb2+(aq) i (aq) pbi2(s) pb(s, pb(s) pb2+(aq) i (aq) pb(s, pb(s) pb2+(aq) i (aq) pbi2(s, pb(s) pb2+(aq) pb(s) pbi2(s) i (aq, none of these. +(aq) + fe2+(aq) n2(g) + fe (s) + 5 h+(aq: n2(g) + 4 ag(s) n2h5, fe2+(aq) + 2 ag(s) fe (s) + 2 ag+(aq) +(aq) + 4 ag+(aq: none of the reactions above is spontaneous. A galvanic cell is constructed from fe/fe2+ and pb/pb2+ half-cells (under standard conditions). Which electrode is the cathode: the fe electrode, the pb electrode, neither (you cannot construct a galvanic cell from these two half-cells) In which direction do positive ions in the salt bridge move: towards fe2+(aq, towards pb2+(aq) N2(g) + 5 h+(aq) + 4e n2h5.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232