CHEM 343 Lecture Notes - Aldehyde, Stereospecificity, Hydroxylation
Document Summary
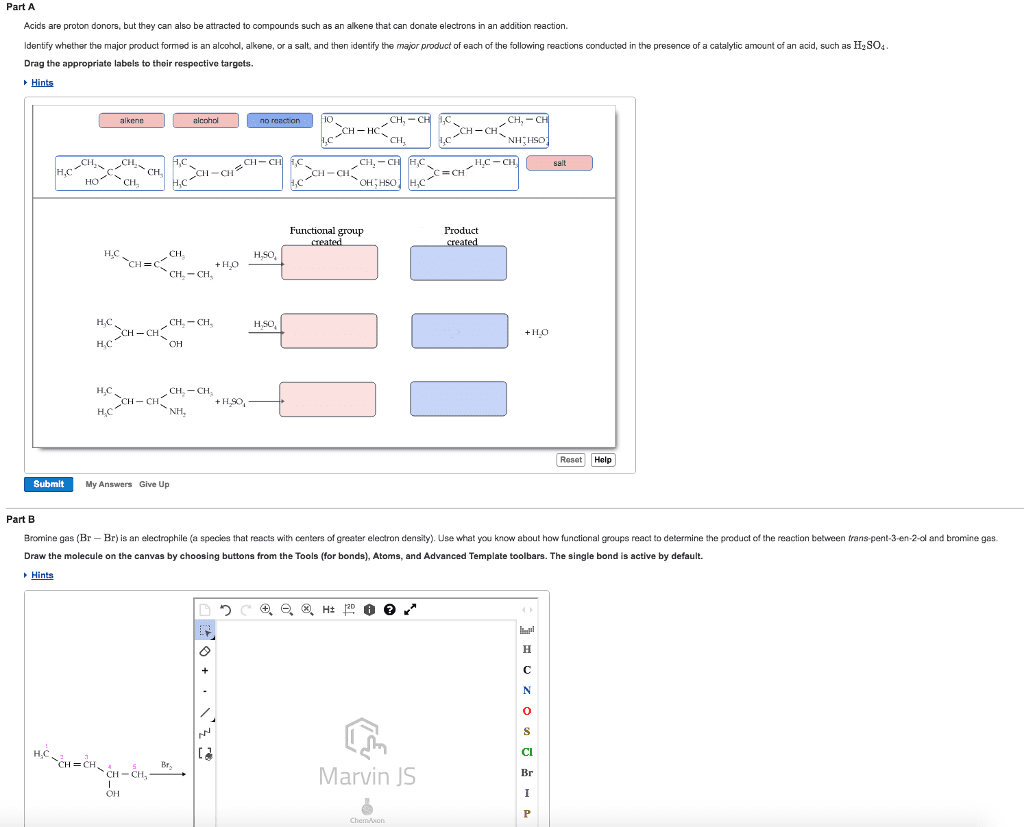
Oxidation loss of electrons (the molecule that loses electrons is oxidized) Reduction gain of electrons (the molecule that gains electrons is reduced) Reduction increases the number of c-h bonds or decreases the number of c-o, c-n, or c-x (halogen) bonds. Oxidation decreases the number of c-h bonds or increases the number of c-o, c-n, or c-x (halogen) bonds. Reduction by addition of two h atoms (catalytic hydrogenation) Reduction by addition of an electron, a proton, an electron, a proton. Dissolving metal reduction metal donates an electron to the compound and ammonia donates a proton. Sodium in liquid ammonia cannot reduce a carbon-carbon double bond. Reduction by addition of a hydride ion and a proton. Lialh4 is stronger reducing agent than nabh4, only lialh4 is strong enough to reduce carboxylic, acids, esters, and amides. Common oxidizing reagents: cro3 (chromium trioxide), h2cro4 (chromic acid), na2cr2o7 (sodium dichromate), and kmno4 (potassium permanganate) Aldehydes are generally easier to oxidize than primary alcohols.