CHEM1101 Study Guide - Midterm Guide: Gibbs Free Energy, Roman Numerals, Manganese

Electro-Chemistry
Electrochemistry is the study of the relationships between electricity and chemical reactions. It
includes the study of both spontaneous and nonspontaneous processes.
Synopsis of Assigning Oxidation Numbers
1. Elements = 0
2. Monatomic ion = charge
3. F: –1
4. O: –2 (unless peroxide = –1)
5. H: +1 (unless a metal hydride = –1)
6. The sum of the oxidation numbers equals the overall charge (0 in a compound).
Oxidation Numbers
• To keep trak of hat loses eletros ad hat gains them, we assign oxidation numbers.
• If the oidatio uer ireases for a eleet, that eleet is oidised.
• If the oidatio uer dereases for a eleet, that eleet is redued
Oxidation and Reduction
• A speies is oidised he it loses electrons. – Zinc loses two
electrons, forming the Zn2+ ion.
• A speies is redued he it gais eletros. – H+ gains an
electron, forming H2.
• A oidisig aget auses soethig else to e oidized H+; a
reducing agent causes something else to be reduced (Zn).
Half-Reactions
• The oidatio ad redutio are ritte ad alaed separatel.
• We ill use the to alae a redo reatio.
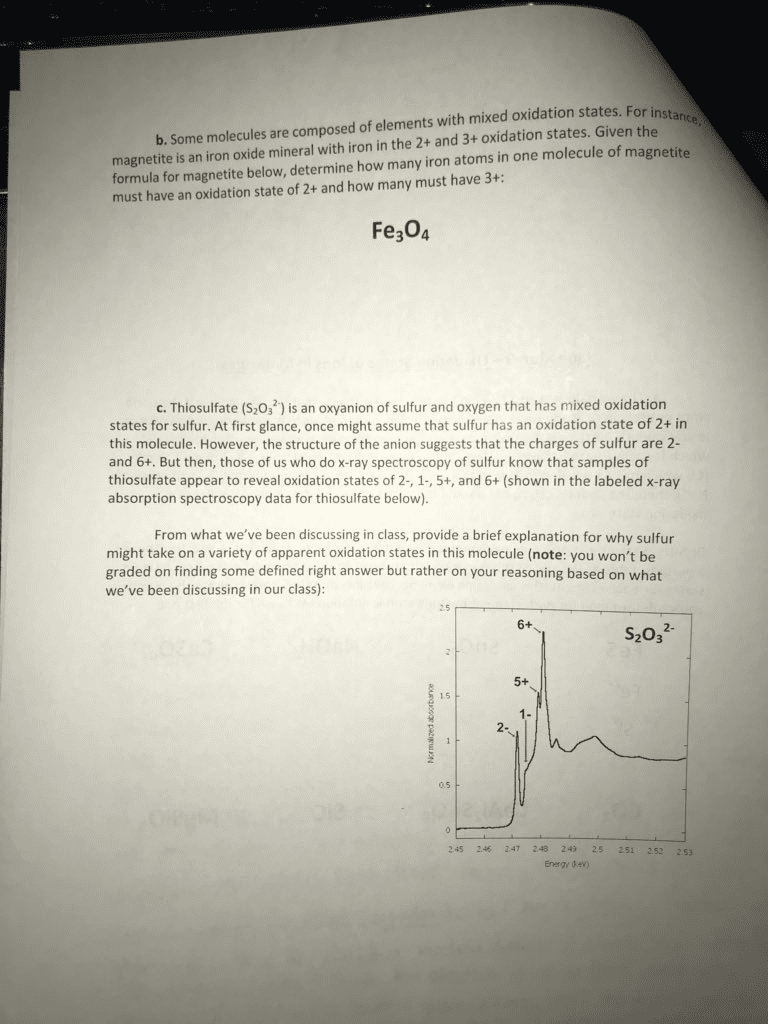
• For eaple, he S+ ad Fe+ reat,
Balancing Redox Equations: The Half-Reactions Method (a Synopsis)
1) Make two half-reactions
(oxidation and reduction).
2) Balance atoms other than
O and H. Then, balance
O and H using H2O/H+.
3) Add electrons to balance charges.
4) Multiply by common factor
to make electrons in half-
reactions equal.
5) Add the half-reactions.
6) Simplify by dividing by common factor or converting H+ to
OH– if basic.
7) Double-check atoms and charges balance!
find more resources at oneclass.com
find more resources at oneclass.com
Document Summary
Electrochemistry is the study of the relationships between electricity and chemical reactions. It includes the study of both spontaneous and nonspontaneous processes. Oxidation and reduction: a spe(cid:272)ies is o(cid:454)idised (cid:449)he(cid:374) it loses electrons. Half-reactions: the o(cid:454)idatio(cid:374) a(cid:374)d redu(cid:272)tio(cid:374) are (cid:449)ritte(cid:374) a(cid:374)d (cid:271)ala(cid:374)(cid:272)ed separatel(cid:455), we (cid:449)ill use the(cid:373) to (cid:271)ala(cid:374)(cid:272)e a redo(cid:454) rea(cid:272)tio(cid:374), for e(cid:454)a(cid:373)ple, (cid:449)he(cid:374) s(cid:374)(cid:1006)+ a(cid:374)d fe(cid:1007)+ rea(cid:272)t, Balancing redox equations: the half-reactions method (a synopsis: make two half-reactions (oxidation and reduction), balance atoms other than. O and h using h2o/h+: add electrons to balance charges, multiply by common factor to make electrons in half- reactions equal, add the half-reactions, simplify by dividing by common factor or converting h+ to. Oh if basic: double-check atoms and charges balance! O= -2 ( x 4) + mg = -(cid:1005), thus mg is + 7 . 2 x 4 atoms + 2c = -2, -8 + 2c = -2 thus c = 4 as the whole thing has to equal -2.