CHEM1101 Study Guide - Midterm Guide: Thermodynamics, Enthalpy

Enthalpy of Reaction
The change in enthalpy, H, is the enthalpy of the products minus the enthalpy of the
reactants:
H = Hproducts − Hreactants
Enthalpy of Reaction This quantity, H, is called the enthalpy of reaction, or the heat of
reaction.
The Truth about Enthalpy
1. Enthalpy is an extensive (depends on the amountof species) property.
2. H for a reaction in the forward direction is equal in size, but opposite in sign, to H for
the reverse reaction.
3. H for a reaction depends on the state of the products and the state of the reactants.
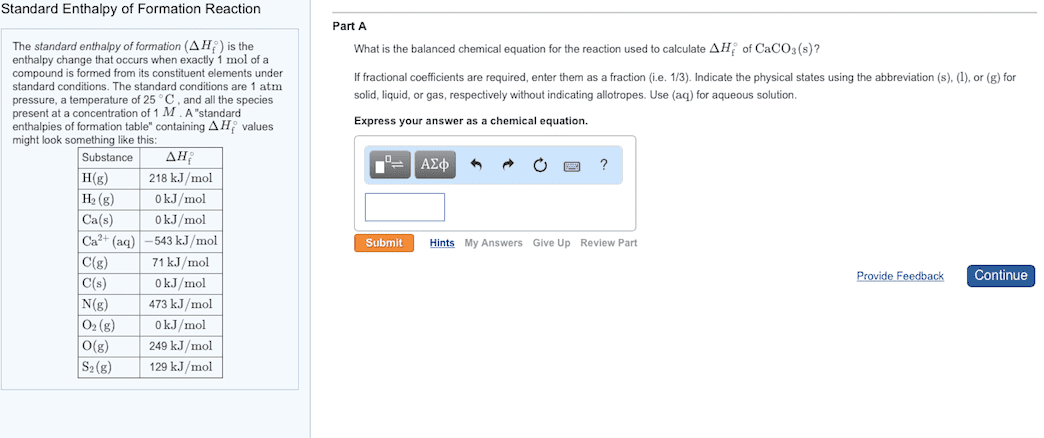
Enthalpies of Formation
An enthalpy of formation, Hf, is defined as the enthalpy change for the reaction in which 1
mol of a compound is made from its constituent elements in their standard state.
Standard Ethalpies of Foratio Stadard ethalpies of foratio, ∆Hf°, are easured
under standard conditions (25 ºC and 1.00 atm pressure).
Calculation of H
C3H8(g) + 5 O2(g) ⎯→ 3 CO2(g) + 4
H2O(l)
Imagine this as occurring in three steps:
1) Decomposition of propane to the
elements:
C3H8(g) ⎯→ 3 C(graphite) + 4 H2(g)
2) Formation of CO2:
3 C(graphite) + 3 O2(g) ⎯→3 CO2(g)
3) Formation of H2O:
4 H2(g) + 2 O2(g) ⎯→ 4 H2O(l)
So, all steps look like this:
C3H8(g) ⎯→ 3 C(graphite) + 4 H2(g)
3 C(graphite) + 3 O2(g) ⎯→3 CO2(g)
4 H2(g) + 2 O2(g) ⎯→ 4 H2O(l)
find more resources at oneclass.com
find more resources at oneclass.com
Document Summary
The change in enthalpy, h, is the enthalpy of the products minus the enthalpy of the reactants: Enthalpy of reaction this quantity, h, is called the enthalpy of reaction, or the heat of reaction. An enthalpy of formation, hf, is defined as the enthalpy change for the reaction in which 1 mol of a compound is made from its constituent elements in their standard state. Standard e(cid:374)thalpies of for(cid:373)atio(cid:374) sta(cid:374)dard e(cid:374)thalpies of for(cid:373)atio(cid:374), hf , are (cid:373)easured under standard conditions (25 c and 1. 00 atm pressure). C3h8(g) + 5 o2(g) 3 co2(g) + 4. Imagine this as occurring in three steps: decomposition of propane to the elements: C3h8(g) 3 c(graphite) + 4 h2(g: formation of co2: 3 c(graphite) + 3 o2(g) 3 co2(g: formation of h2o: 4 h2(g) + 2 o2(g) 4 h2o(l) The sum of these equations is the overall equation! C3h8(g) + 5 o2(g) 3 co2(g) + 4 h2o(l)