
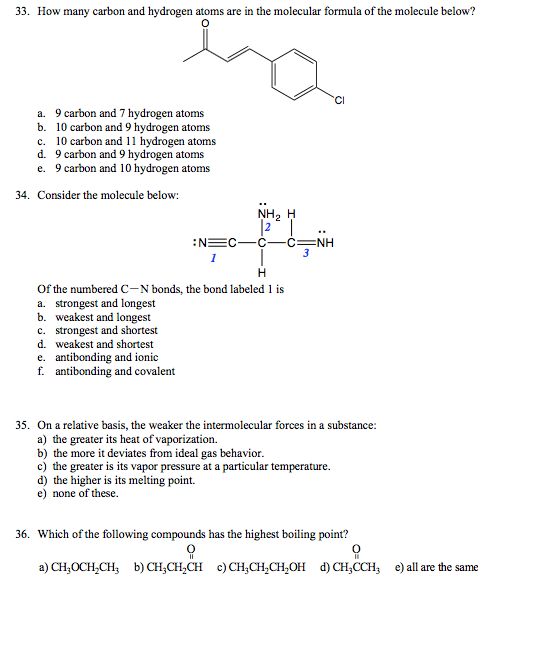

28. Which is the best Lewis structure for acetyl chloride, CH 3COO)CI? b) H :O: c) H :O: c) H :O: 29. A resonance form for the thiocyanate ion, NCs, has the bonding pattern shown below N C S Complete the Lewis structure. The formal charges on N, and S, respectively, are a) +2, 0,-1 b) 0, 0,-1 c) -2, 0, +1 d) -1, 0, 0 e) -2, +4, -1 30. Which of the following molecules are polar? C O a) 1 b) 2,3,4 c) 2,4 d all of them e) none of them 31. Choose the correct statement about the phase diagram below. 1 atm -f a) The diagram is qualitatively correct for water. b) The diagram shows that the melting point of the solid increases with increasing pressure c) The diagram shows the triple point above 1 atm pressure. d) The diagram could represent the phase diagram for CO2. e) None of the above statements are correct. 32. Which of the following represents a possible set of quantum numbers for an electron in an atom? a) n 3, 0, m 1, m 1/2 b) n 3, 1-2, mi 2, m 1/2 c) n 3, 3, m -1, ms -1/2 d) n 3, l 2, m 0, m 0 e) n 3, 0, m 1, m -1/2 none of the sets are possible






