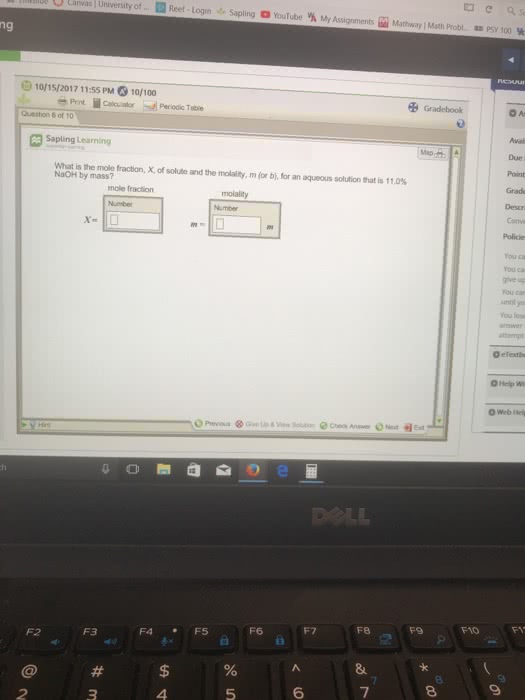
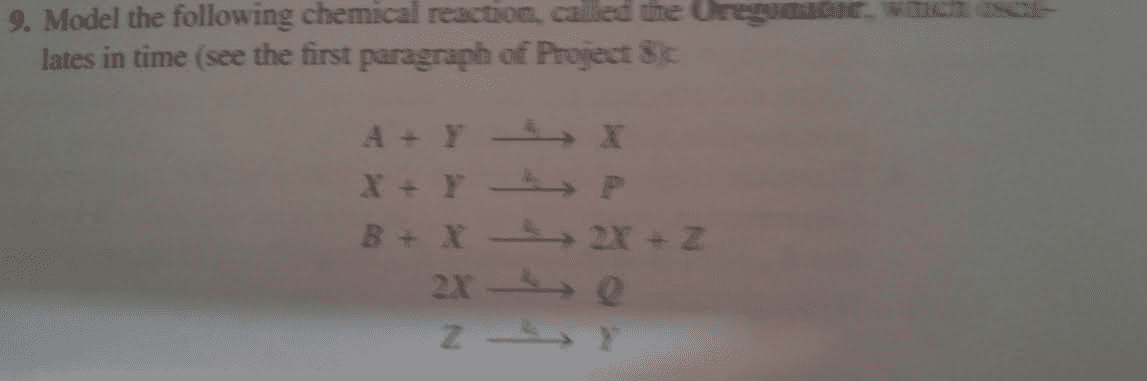CHEM 3503 Midterm: Exam-CHEM3503-2002December
Document Summary
D u r a t i o n : 3 h o - u r s. N o , o f s t u c j e n t s : 7 5 f " \ o n a r t m o n i u c l j a : l. C l t a r n i 5 t " y 6 5 " 3 5 3. C n r r r c o l n a t " r , c t r l / / q l r l t. I describe the pattern of atomic properties obsen"ed when elements are put in order of increasing atomic weight. Erplain how this pattern was reconstructed into the long form of the periodic table. 1 what two kev exoeriments showed that atoms were divisible ? tifr t- J define the range of values and the structural aspects of orbitals are identified by quantum numbers n, i and m".