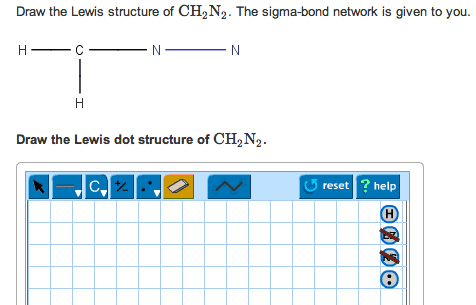
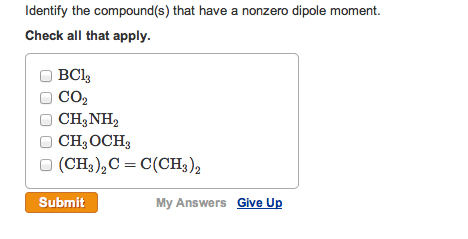
CH2610 Quiz: Solution CH2610 Assignment 2
Document Summary
Chemistry 2610 assignment 2: questions and solutions. Name: __________________: consider ethane, c2h6, and do the following: Identify the shapes and bond angles of ethane. H tetrahedral about each carbon atom bond angles are ~ 109. 5o carbon atoms are sp3 hybridized. Identify the hybridization of the two carbon atoms, and draw the overlapping atomic orbitals of the carbon and hydrogen atoms, and thereby identify the bonding molecular orbitals of ethane. the c-c bond is sp3-sp3 = (sigma) bond. 6 x h-c bonds are s-sp3 = (sigma) bond. 1: consider ethene, c2h4, and do the following: Identify the shapes and bond angles of ethene. H trigonal planar about each carbon atom bond angles are ~ 120o carbon atoms are sp2 hybridized. Identify the hybridization of the two carbon atoms, and draw the overlapping atomic orbitals of the carbon and hydrogen atoms, and thereby identify the bonding molecular orbitals of ethene. (pi) bond the c-c bond is sp2-sp2 = (sigma) bond.