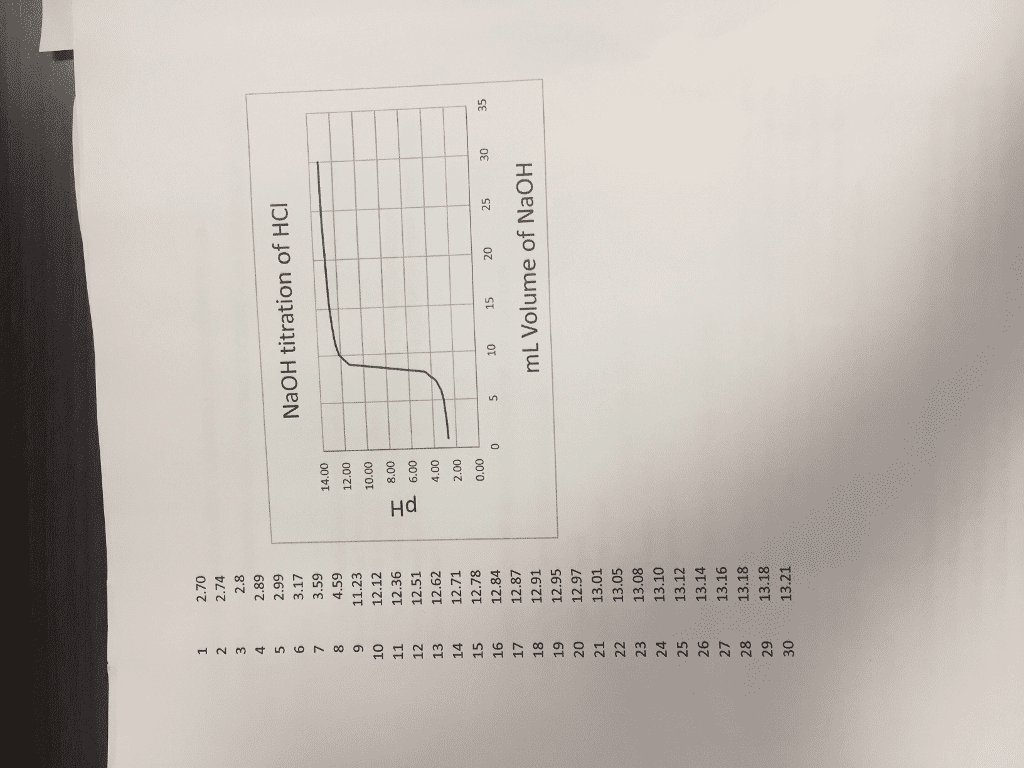
CHEM123L Study Guide - Final Guide: Magnetic Stirrer, Purified Water, Ph Meter
273 views2 pages
18 Mar 2014
School
Department
Course
Professor
Document Summary
Experiment procedure summary 4: ph chemistry: acid base titrations and buffers: sign out from storeroom, 1 50 ml pipette bulb, 1 50 ml burette, 1 magnetic stirring bar, 1 25 ml pipette. Make sure that your ph meter has been calibrated. The ph meter should be turned on and the numbers 7 and 4 should be showing. Part a: titrations of a weak acid with strong base. Pipette 25 ml of 0. 1 m acetic acid into a clean 400 ml beaker by placing the magnetic bar first. Copy the exact molarity given on the stock bottle. Add 150 ml deionized water and 2 drops of phenolphthalein. Obtain 60 ml of the unknown naoh solution and prepare for titration. Place it in position to titrate to ch3cooh solution. Record the burette readings and ph, both to two decimals place. Part b: titrations of a weak base with a strong acid. Pipette 25 ml of 0. 1m ammonia into a clean 400 ml.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232