CHEM 313 Study Guide - Midterm Guide: Stereochemistry, Sodium Hydroxide, Covalent Bond
Document Summary
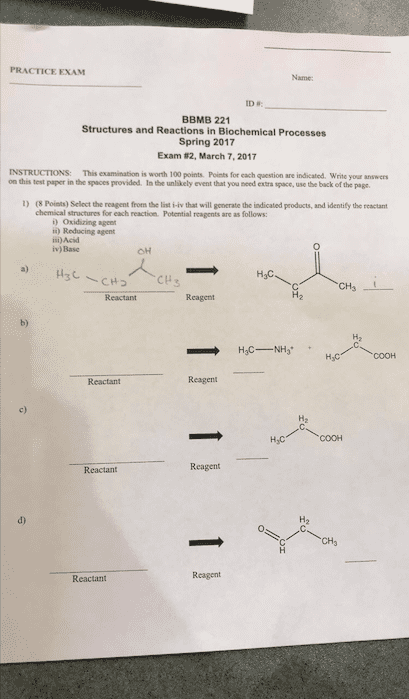
7 october 2013 (this exam is out of 100 points. ) [35 points] predict the products of the reactions below. Nh2 (b) calculate the pi (isoelectric point) of cysteine. [8 points] (a) draw the predominant structure of l-cysteine at ph 5. [7 points] calculate the equilibrium constant for the reaction below (show your work): [10 points] (a) the compound below exists as an ionic compound. Draw a structure of it that better represents its ionic nature. Br (b) rationalize why this compound is ionic rather than covalent: 3. [20 points] (a) propose a mechanism for the reaction below. Ch2 (b) propose an alternative (constitutional) isomeric product. (c) rationalize why little or none of the isomer you have drawn in part (b) is isolated: 4. [20 points] draw resonance structures of diazomethane, ch2n2 (note: the connection is c-n-n not n-c-n, nor is it cyclic). Rank your resonance structures in terms of their relative weightings, and explain your rankings: 5.