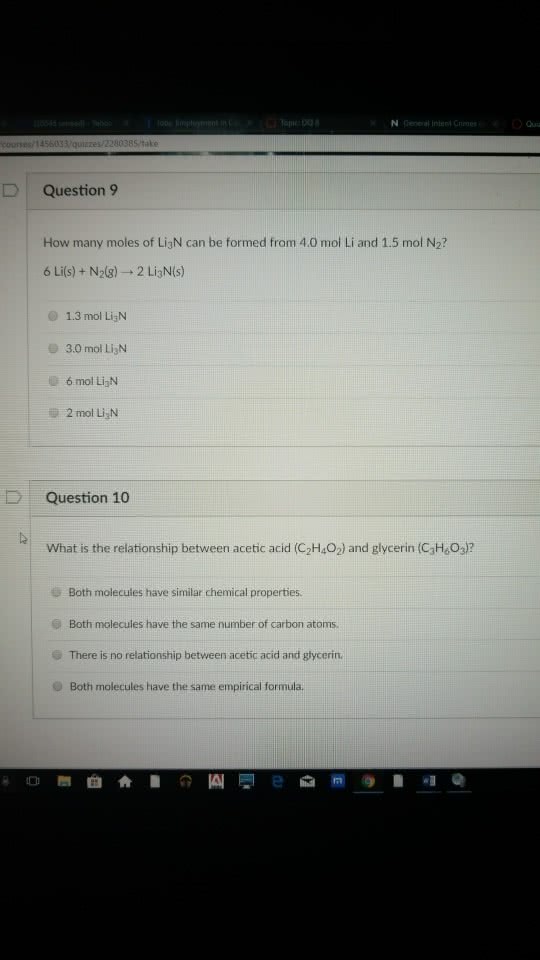
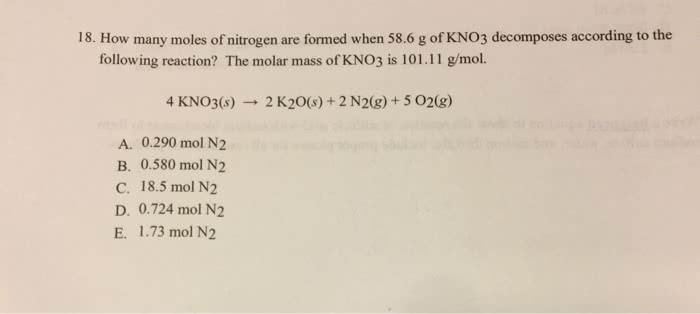lopic Da N General Intent Crime Quis course,/1456033/quizzes/2280 ake DQuestion 9 How many moles of LigN can be formed from 4.0 mol Li and 1.5 mol N2? 6 Li(s) + N2(g) â 2 Li3N(s) 1.3 mol LigN 3.0 mol Li3N 6mol Li3N e 2 mol LigN DQuestion 10 What is the relationship between acetic acid (C2H4O2) and glycerin (C3H&O3) Both molecules have similar chemical properties. O Both molecules have the same number of carbon atoms O There is no relationship between acetic acid and glycerin. 0 Both molecules have the same empirical formula. 0å³æ²ì 1ì ë¡ eå §[email protected]