CHEM 102 Midterm: EXAM I REVIEW
137 views5 pages
1 Feb 2017
School
Department
Course
Professor
Document Summary
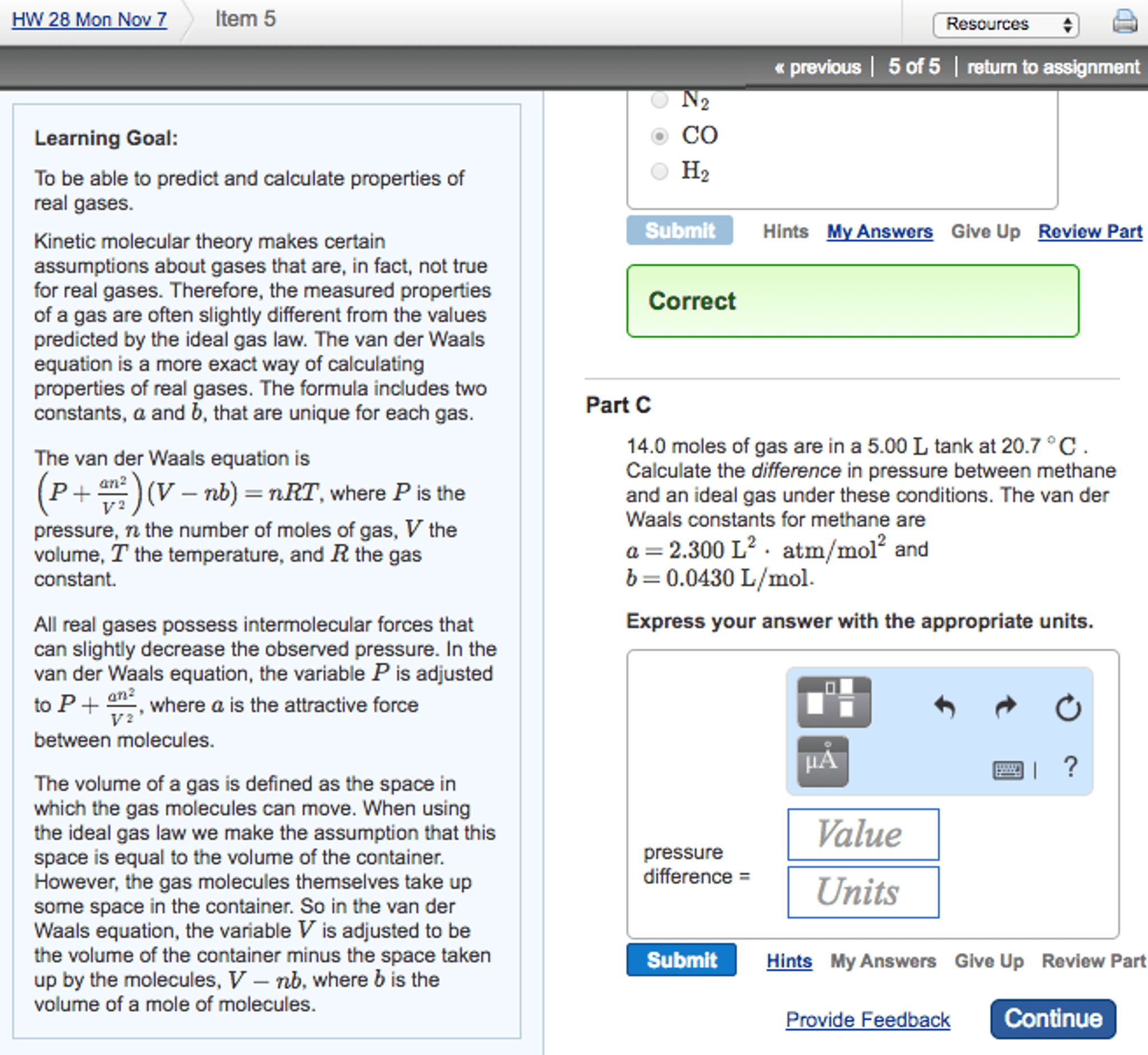
Greatest deviation from ideal gas law happens at low temperature and high pressure. (cid:1853)/2=adjusts p for the molecular attraction (pressure is increased) Note: never will see a higher pressure than in ideal gas law (real gases have lower ps) Polar molecules have a higher bp than nonpolar molecules. Larger and polar molecules have more and/or stronger interactions=higher. Attractions between positive and negative ends of neighboring molecules. Polar molecules have more/stronger interactions with partially positive and partially negative attractions. H bonded directly to an extremely electronegative atom (typically o, n or f) and its interaction is with a lone pair of electrons. Dispersion forces < dipole-dipole < hydrogen < ion-dipole. Qualitative term for the tendency of a liquid to vaporize. Quantitative term for the pressure of the vapor above the surface of a liquid f o l s e u c e o m l. Bp is reached when vp=the atmospheric pressure.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232