CHEM 330 Study Guide - Quiz Guide: Infrared Spectroscopy, Human Development Index, Wavenumber
Document Summary
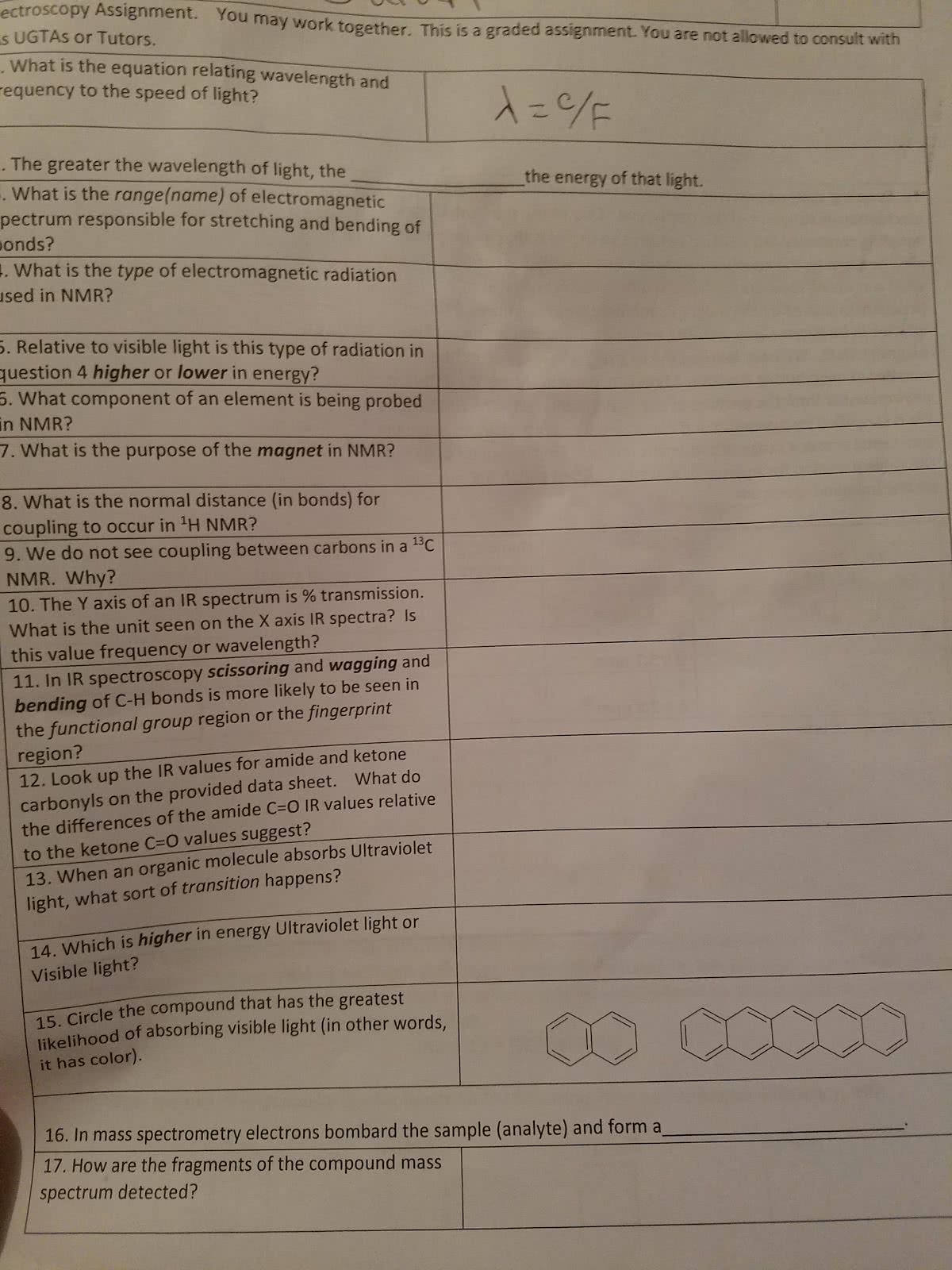
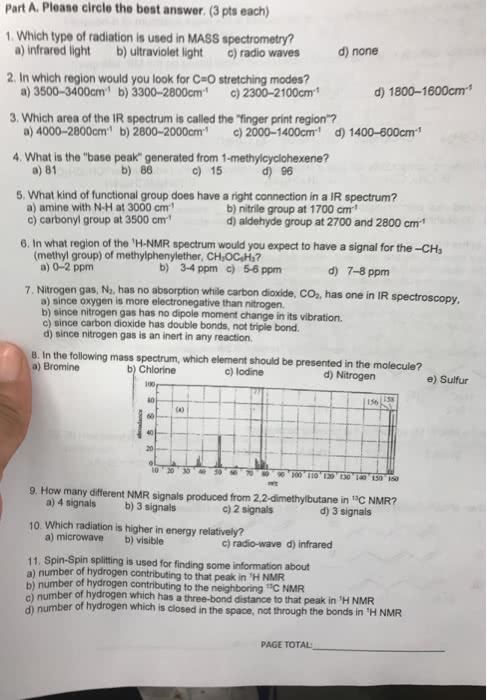
Electromagnetic radiation can be exhibited as a wave or particle. Ir spectroscopy tells us the functional groups present in the compound. Promotes vibrational excitations of the bonds in a molecule. C-h bonds absorb higher radiation and will have that radiation characteristic. Larger wavenumber means higher energy, shorter wavelength, higher frequency f= c/l. As shown, the carbonyl group of an unsaturated, conjugated ketone produces a signal at lowe wavenumber (1680 cm 1) C-h bonds have large intensities and absorb higher radiation. Vibration modes with a few atoms can be used to identify functional groups. Asymmetric and bending bonds are shown as peaks. The smaller the reduced mass the higher the stretching frequency. The r-group could be any size alkane side group that contains fewer carbons than the paren carbon chain. Knowing the hdi formula and how to use it: always integer. Tells you how many molecules of hydrogen you need to saturate.