Theoretical and percent yield problems
1. Arcylonitrile C3H3N is the starting material for the production of a kind of synthetic fiber and can be made by a chemical reaction between propylene C3H6 and nitric oxide NO. The unbalanced reaction is below. What mass of C3H3N can be made when 21.6 g of C3H6 react with 21.6 g of nitric oxide? Identify the limiting reagent and what theoretical yield will be. If 24.3 g of C3H3N are recovered, what is the percent yield? First balance the chemical reaction.
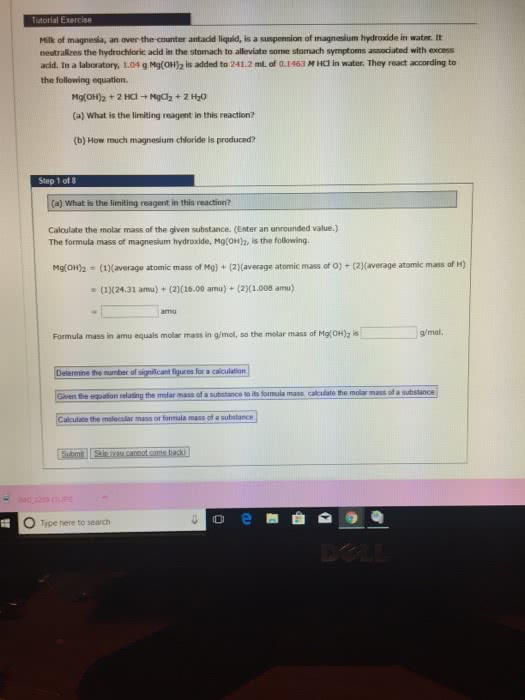
2. A 50.6 g sample of Mg(OH)2 is readers with a 45 g of HCl according to the reaction. What is the limiting reagent and what is the theoretical yield of MgCl2 are recovered what percent yield? First balance the equation.
3. If you start with ten grams or lithium hydroxide, how many grams of lithium bromide will be produced if excess HBr is used?

Theoretical and pereent yield problems 1. Arcylonitrile, CH,N, is the starting material for the production of a kind of synthetic fiber and can be made by a chemical reaction between propylene CiHs and nitric oxide NO. The UNBALANCED reaction is below. What mass of C.H&N can be made when 21.6 g of CHs, react with 21.6 g of nitric oxide? Identify the limiting reagent and what the theoretical yield will be. If 24.3 g of CHN are recovered, what is the percent yield? First balance the chemical reaction. C,H, (g) C,H,N (s) + H2O (l) + N2 (g) + NO (g) â A 50.6 g sample of Mg(OH)2 is reacted with 45.0 g of HCl according to the reaction. What is the limiting reagent and what is the theoretical yield of MgClh? If 55.7 g of MgCly are recovered what is the percent yield? First 2. balance the reaction. HCI MgCl2 + H2O â Mg(OH)2 + 3. If you start with ten grams of lithium hydroxide, how many grams of lithium bromide will be produced if excess HBr is used? LiOH + HBr â LiBr + H2O