CHEM 1LD Study Guide - Final Guide: Volumetric Flask, Intermolecular Force, Molar Mass
383 views5 pages
Document Summary
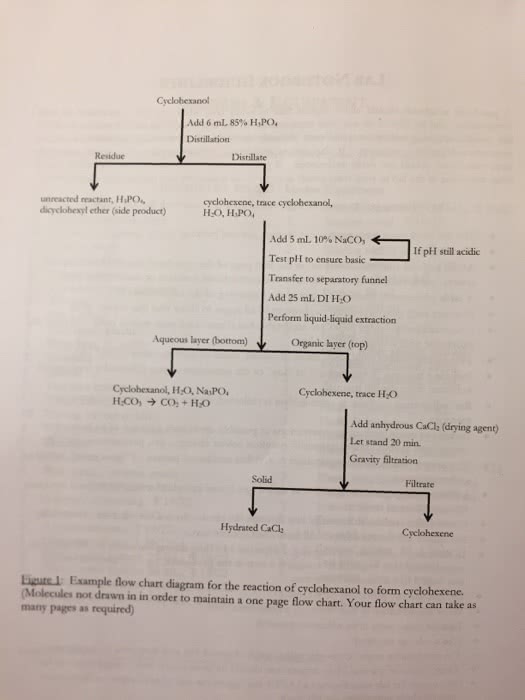
Density of mixture in the 10 ml vol flask mass of mixture in the. Calculate the percent change in volume for the mixture. As forces increase the volume decreases making the change in volume negative. Which means the more force pull and attract each other the lesser the volume will be. H/r is the slope if you look a it in a y=mx+b so to find h get the slope using y2- y2/x2-x1 for two points then you will divide by the r constant. Multiply the top reaction by 2 and the bottom one by 5 to make the number of electrons gained and lost the same. Other question: balanced equation for iron salt show half reactions. 2 fe(nh4)2(so4)2 6h2o + _3__ h2c2o4 + _3_ k2c2o4 + _1_ h2o2 __2_ k3fe(c2o4)3 3h2o + 4 spots measure from origin to the spot then from the origin to the top solvent line .
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232