CHEM 1124Q Study Guide - Midterm Guide: Dinitrogen Pentoxide, Aluminium Oxide, Main-Group Element
Document Summary
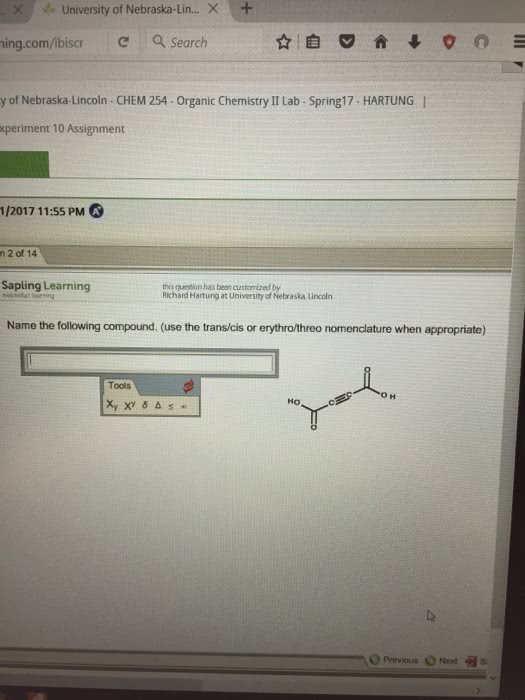
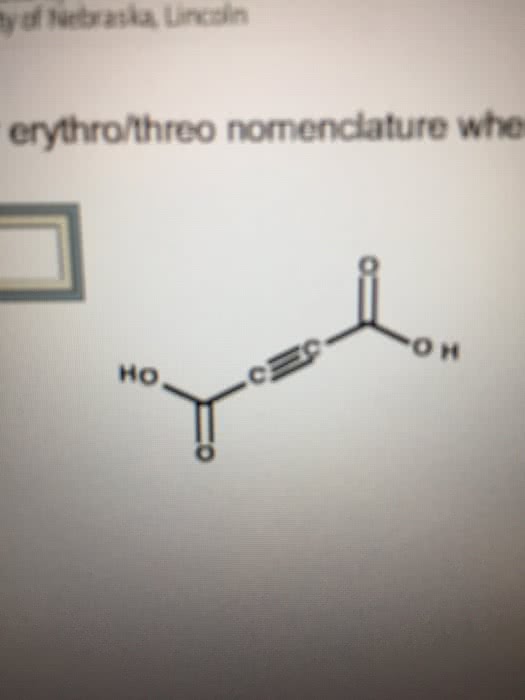
Naming ions and binary ionic compounds 13. Polyatomic ions - oxoanions and oxoacids 18. Dipole moment, partial charge and percent ionic character 27. Lewis dot structures and formal charge 28. Molecular geometry & the vsepr model, intermolecular forces, and bonding theories 30. Geometry of molecules with more than one central atom 32. Van der waal forces and hybridization 34. In 1864, john newlands noted that when the elements were arranged in order of atomic mass that every eighth element had similar properties: he referred to this as the law of octaves. In 1869, dmitri mendeleev and lothar meyer independently proposed the idea of periodicity. Mendeleev grouped elements (66) according to properties. Mendeleev predicted properties for elements not yet discovered, such as ga. Mendeleev could not explain inconsistencies such as tellurium coming before iodine in the periodic table, despite having a higher atomic mass. Method of discovery probably from its spectrum spectroscopically.