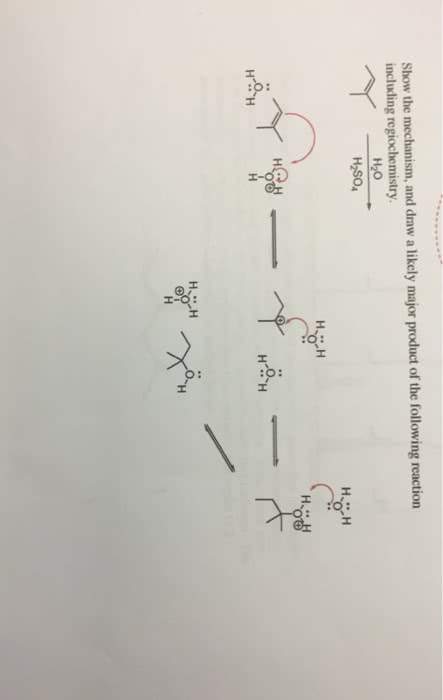
Chem 2 lab test tomorrow this is study guide
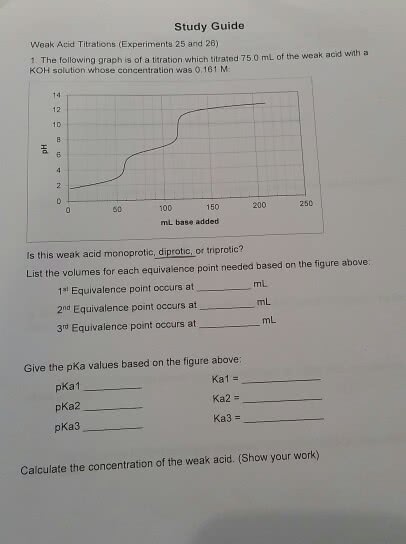
Study Guide Weak Acid Titrations (Experiments 25 and 26) 1. The following graph is of a tration wh ch titrated 75.0 mL of the weak acid with a KOH solution whose concentration was 0,161 M 1D 50 100 150 250 mL base added Is this weak acid monoprotic, diprotic, or triprotic? List the volumes for each equivalence point needed based on the figure above 1s Equivalence point occurs at 2nd Equivalence point occurs at 3rd Equivalence point occurs at mL mL mL Give the pKa values based on the figure above: pKa1 pKa2 pKa3 Kal = Ka2 = Ka3 = Calculate the concentration of the weak acid. (Show your work)