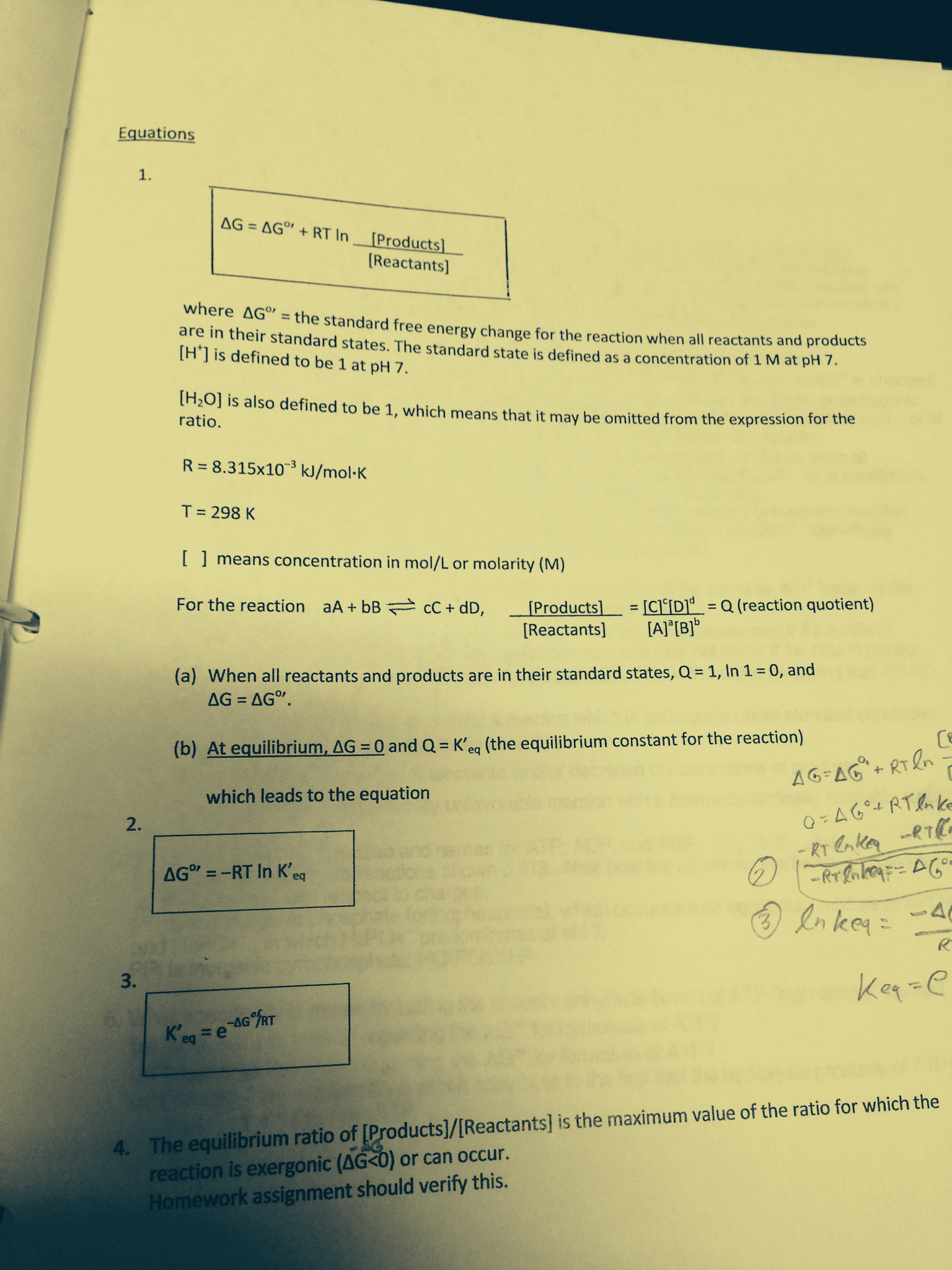University College - Chemistry Chem 112A Midterm: Sp18 Exam 3 Study Guide
57 views12 pages
14 Aug 2018
School
Department
Professor
Document Summary
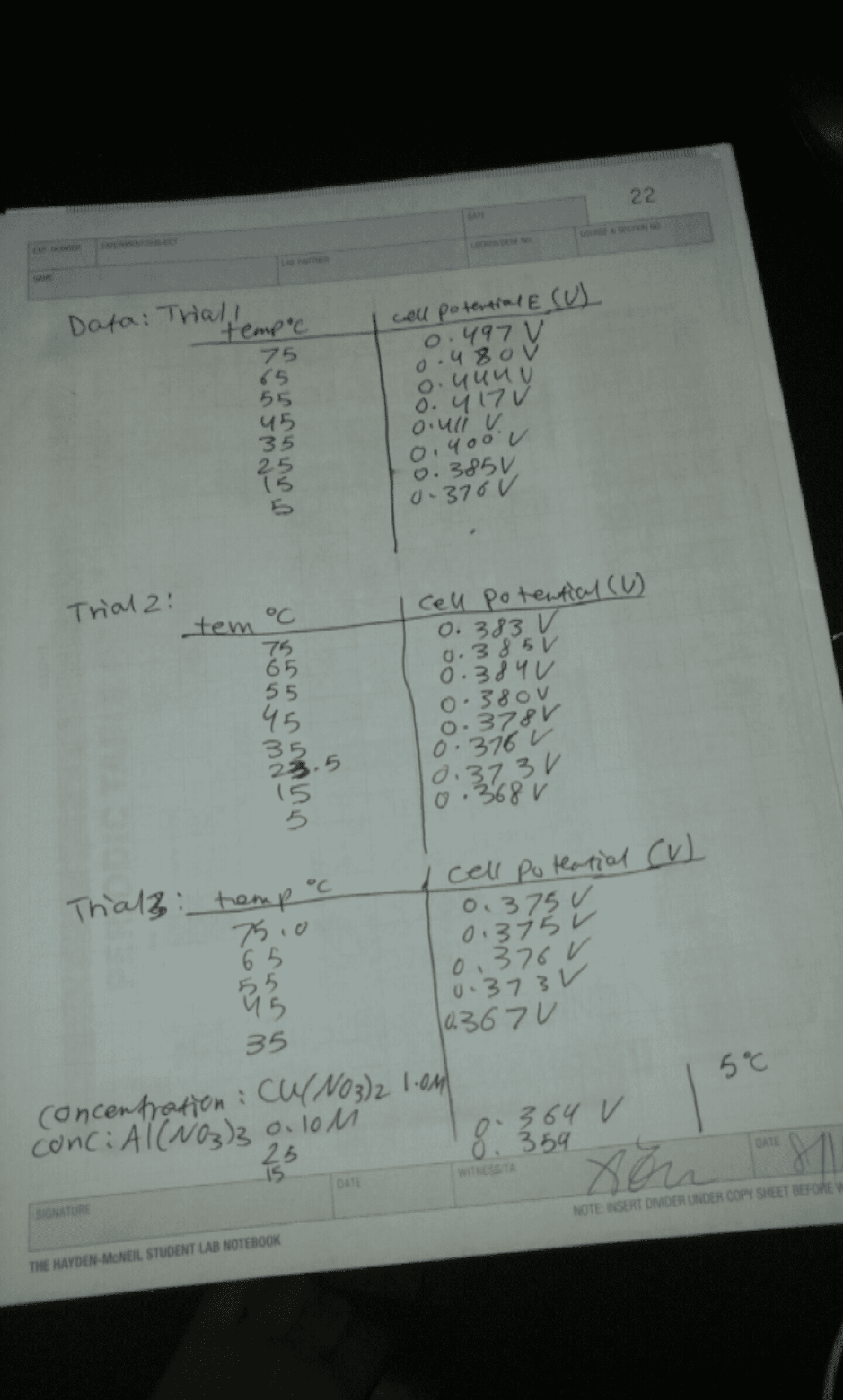
Decreasing: proves that in an exothermic reaction the higher the t the lower the k, proves that in an endothermic reaction the lower the t the higher the k, assumes that ho and so are temperature independent. Calculation of total free energy for the reaction a (g) b (g): at every point along the reaction coordinate, you can calculate, ga, gb, and g total, ga = go. A + rt lnp a (kj/mol: gb = go. B + rt lnp b (kj/mol: g tot = n a ga + n b gb = n a(go, as the reaction proceeds, a (g) is converted to b(g), so at every point along the reaction. A + rt lnp a) + n b (go. A + rt ln(n a p total)] + n b [go. A + rt ln(n a p total)] +(1- n a)[go. Introduction to electrochemistry: electrochemistry is the study of inter conversion of chemical and electrical chemistry.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232