CHEM 2OA3 Chapter Notes - Chapter 1: Orbital Hybridisation, Pauli Exclusion Principle, Statcoulomb
Document Summary
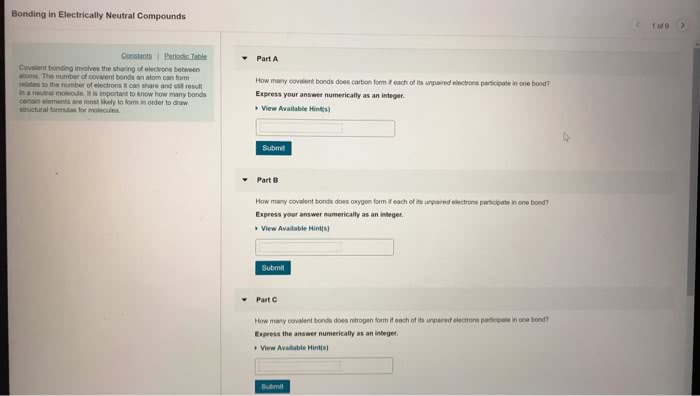
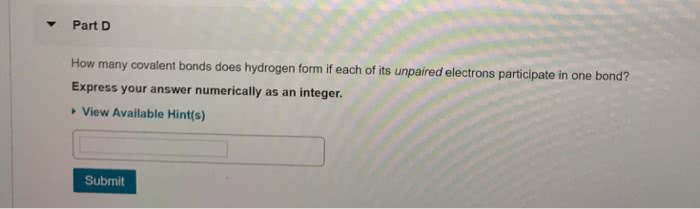
Three individuals suggested that substances are defined by a specific arrangement of atoms. Structures of ammonium cyanate and urea: both have same molecular formula (ch4n2o), but differ from each other in way atoms are connected. Are therefore called constitutional isomers: have different physical properties and different names. According to structural theory of matter, each element will generally form a predictable number of bonds. Valence describes number of bonds usually formed by each element: ex. Carbon generally forms four bonds and is therefore said to be tetravalent. A covalent bond can be defined as the result of two atoms sharing a pair of electrons: ex. When these electrons are shared to form a bond, there is a decrease in energy, indicated by negative value of h. As atoms get closer together, forces become stronger. Electrons are able to move to minimize repulsive forces and maximize attractive forces. Provides a net force of attraction, which lowers energy of system.