ENV100Y5 Chapter Notes - Chapter 2: Mid-Atlantic Ridge, Seismic Wave, Soil Chemistry

69
ENV100Y5 Full Course Notes
Verified Note
69 documents
Document Summary
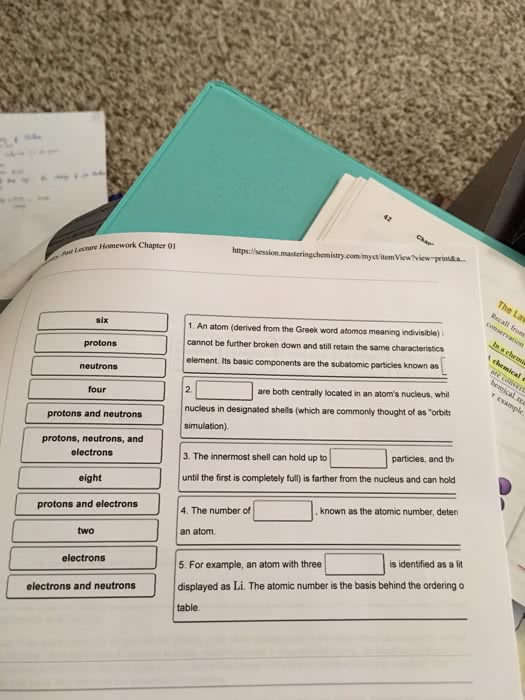
Chapter 2- matter, energy, and the physical environment. Matter- any material that has mass and occupies space. Law of conservation of matter: matter may be transformed from one type of substance into others, but it cannot be created or destroyed. Elements- a fundamental type of matter; a chemical substance with a given set of properties, which cannot be broken down into substances with other properties. Atoms- the smallest component of an element that maintains the chemical properties of that element which consists of: Atomic number: atoms of each element have defined number of protons. Isotopes- one of several forms of an element having differing numbers of neutrons in the nucleus of its atoms. Ions- an electrically charged atom or combination of atoms. Cations: ions that form when an atom loses electrons, and thus, carry a positive charge. Anions: ions that form when an atom gains electrons, and thus, carry a negative charge. Molecules- a combination of two or more atoms.