CHM136H1 Chapter Notes - Chapter 1.3 - 1.4: Noble Gas, Ion, Pauli Exclusion Principle

34
CHM136H1 Full Course Notes
Verified Note
34 documents
Document Summary
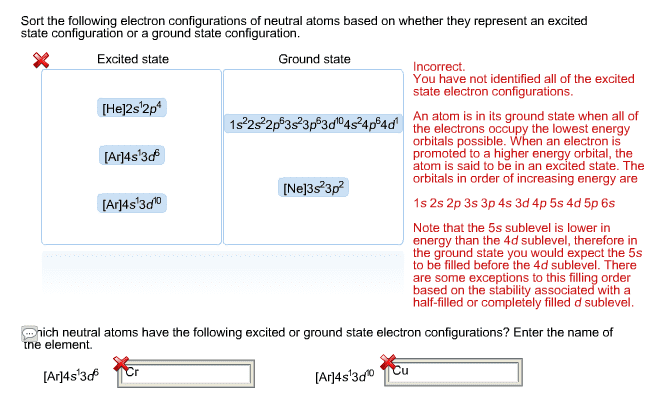
The lowest-energy arrangement, or ground-state electron configuration, of an atom is a listing of the orbitals occupied by its electrons. This spin can have two orientations, denoted as up ( ) and down ( ). Only two electrons can occupy an orbital, and they must be of opposite spin, a statement called the pauli exclusion. Principle: if two or more empty orbitals of equal energy are available, one electron occupies each with spins parallel until all orbitals are half-full, a statement called hund"s rule. Hydrogen, for instance, has only one electron, which must occupy the lowest-energy orbital: thus, hydrogen has a 1s ground-state configuration. Carbon has six electrons and the ground-state configuration 1s2. A superscript is used to represent the number of electrons in a particular orbit. In 1865, kekul provided another major advance when he suggested that carbon chains can double back on themselves to form rings of atoms. Atoms bond together because the compound that results is more.