Chemistry 1301A/B Chapter Notes - Chapter 3.1: Trigonal Planar Molecular Geometry, Coordination Sphere, Coordination Complex
Document Summary
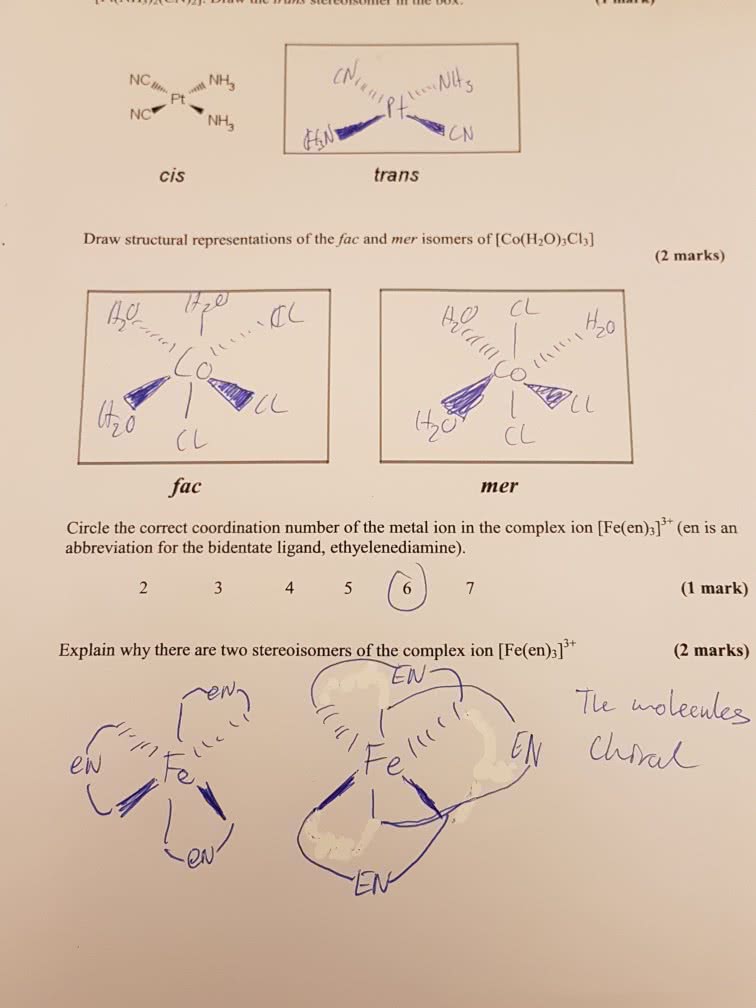
Coordination isomerism: a form of structural isomerism in which the composition of the complex ion varies, the total ratio of ligand to metal remains the same but the ligands attached to a specific metal ion change. It is only possible for salts in which both the cation and the anion are complex ions. Coordination numbers and stereochemistry: the primary term to describe these complexes is coordination number, which is the number of atoms directly bonded to the metal center. In order to get a neutral complex with these metal ions, one anionic ligand and one or two neutral ligands are needed. Coordination number 4: stereochemistry can be tetrahedral or square planar, to form neutral complexes, two neutral ligands and two anionic ligands are required, for platinum, there are two possible isomers in which the angles of x-pt-x can be. 180 degrees (trans isomer) or 90 degrees (cis isomer: these stereoisomers have different chemical and physical properties.