CAS CH 102 Chapter Notes - Chapter 2: Chlorine, Weighted Arithmetic Mean, Niels Bohr
Document Summary
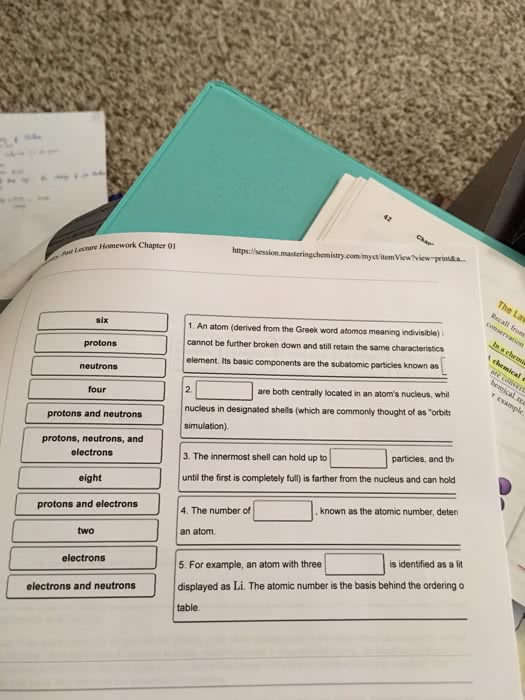
Chapter 2: the composition and structure of the atom. 2. 1 matter and structure: understanding the structure of the atom will help to understand the properties of the elements, keep in mind that these, as all theories, are subject to constant refinement. 2. 2 composition of the atom: atom the basic structural unit of an element. Neutrons: nucleus small, dense, positively charged region in the center of the atom. Neutrons uncharged particles: surrounding the nucleus is a diffuse region of negative charge populated by: Electrons negatively charged particles: atomic number the number of protons in an atom, mass number sum of the number of protons and neutrons, isotopes atoms of the same element having different masses. Chlorine consists of chlorine-35 and chlorine-37 in a 3:1 ratio. Calculate the atomic mass of naturally occurring chlorine if 75. 77% of chlorine atoms are chlorine-35 and 24. 23% of chlorine atoms are chlorine- Step 1: convert the percentage to a decimal fraction.