CHEM 140 Chapter Notes - Chapter 9: Osmosis, Molar Concentration, Volume Fraction
77 views4 pages
30 Apr 2017
School
Department
Course
Professor
Document Summary
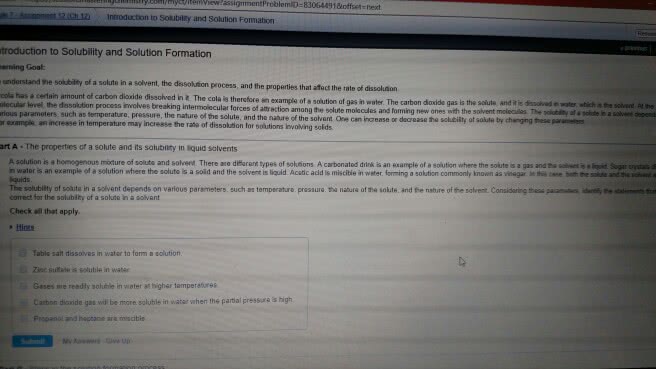
A solution is a homogeneous mixture where the solute is uniformly distributed in a substance called the solvent. Particles of the solute are evenly dispersed among the molecules of the solvent. The solution that forms is in the same physical state as the solvent. Water is known as the universal solvent and is very common in nature. O-h bond is very polar, water is a polar solvent because the o atom has a partial negative charge and the h atom has a partial negative change. Hydrogen bonds occur between molecules where partially positive hydrogen atoms are attracted to partially negative atoms. Interactions between solute and solvent determine whether a solution will form. Energy is needed to separate solute and solvent particles. Energy is released as solute particles move in the solvent to form a solution. Attraction accounts for the first separation and this will occur when solute and solvent have similar polarities- like dissolves like.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232