CHEM 1201 Chapter : Chapter 3 Notes
Document Summary
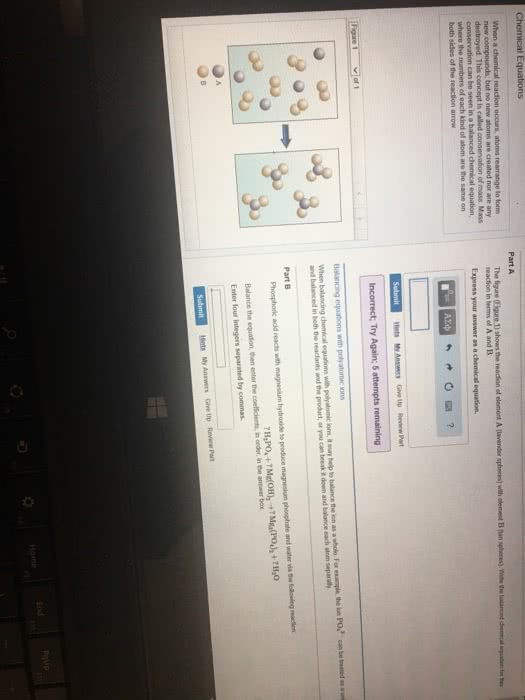
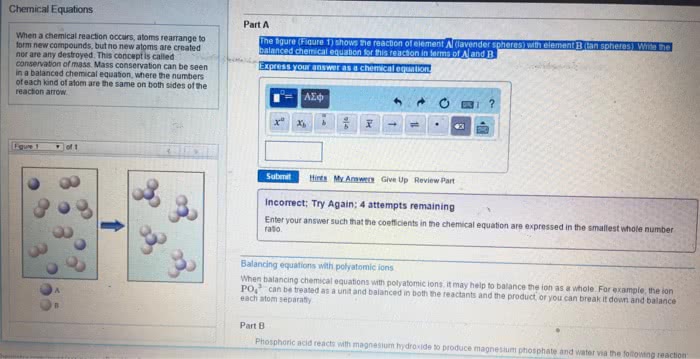
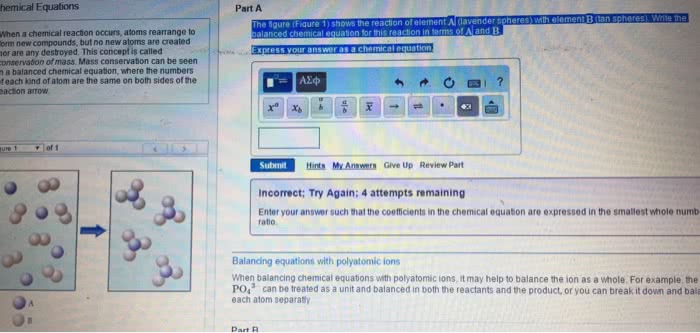
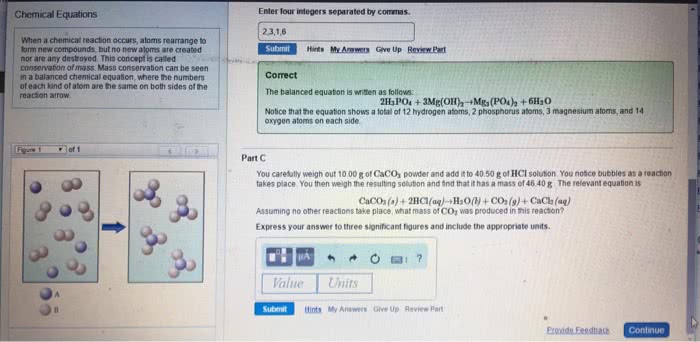
3. 1 chemical equations: the quantitative nature of chemical formulas and reactions is called stoichiometry, lavoisier observed that mass is conserved in a chemical reaction, this observation is known as the law of conservation of mass. Indicating the states of reactants and products: the physical state of each reactant and product may be added to the equation: Ch4(g) + 2o2(g) co2(g) + 2h2o(g: reaction conditions occasionally appear above or below the reaction arrow (e. g. , " " is often used to indicate the addition of heat). Forward references: stoichiometric coefficients will be used to determine molar ratios (stoichiometric factors) in stoichiometric questions later in chapter 3 as well as in chapter 4 (section. 4. 6 on solution stoichiometry), chapter 5 (stoichiometry of heat and hess"s law), chapter. Combination and decomposition reactions: in combination reactions two or more substances react to form one product, combination reactions have more reactants than products, consider the reaction: