CHEM 1201 Chapter : Chapter 5 Quiz 1 (with Answers)
8 views2 pages
Document Summary
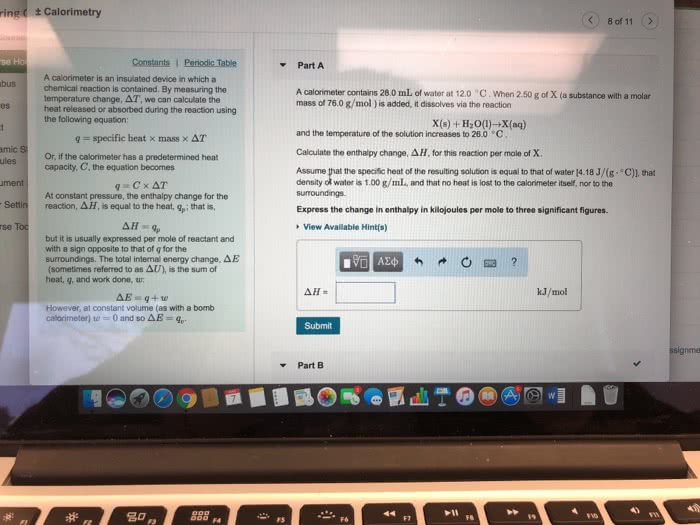
Inserting the numbers from this problem the equation becomes. Note that the sign of the work is negative because the words of the problem say the work was done on the surroundings. Therefor the sign of the work is negative. Solving for heat we obtain the result q 367. In this case we are given the mass and we need to find the heat generated: 0 g na, 99 g/mol. This is used to convert grams to moles. During the solid to gas phase transition energy is required to separate the molecules. Energy must be added to the system so that the molecules can overcome the intermolecular forces that hold the solid together. At constant pressure q h: to understand what work is being done remember that work is a force moving through a distance. A solid ocupies a small volume compared to a gas.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232