CHEM 1202 Chapter : Chapter 16 Exam Review Notes
Document Summary
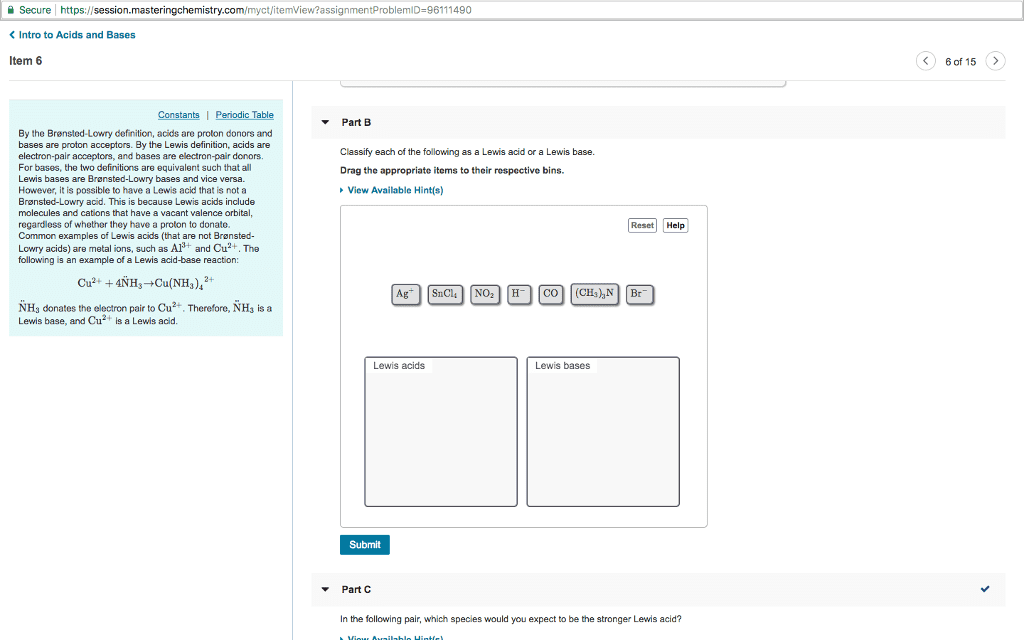
We call the products conjugate acids and bases: acid-base strength, the concept of acid or base strength is related to the amount of reaction. The stronger the acid is the weaker the conjugate base is: the weaker the acid the less it dissociates into ions. [oh-]=10-poh: pka and pkb, the equilibrium constants for acids and bases, ka and kb can also be expressed using a base 10 log scale. pk equations pka=-log[ka] pkb=-log[kb] Kw=[h+][oh-]=1. 0x10-14 at 298 k in water pkw=-log[kw]=ph+poh=+14. 00: polyprotic acids, acids that can dissociate to yield more than one proton are called polyprotic acids. In most cases the dissociation of the second proton is negligible compared to the first. It is much easier to remove the first proton: 2nd step is usually negligible compared to the 1st step, weak acids and bases, they do not dissociate completely, equilibrium lies far on the reactant side kc<<1, checking approximations.