CHEM 2261 Chapter : Notes2261Chapter6
12 views9 pages
Document Summary
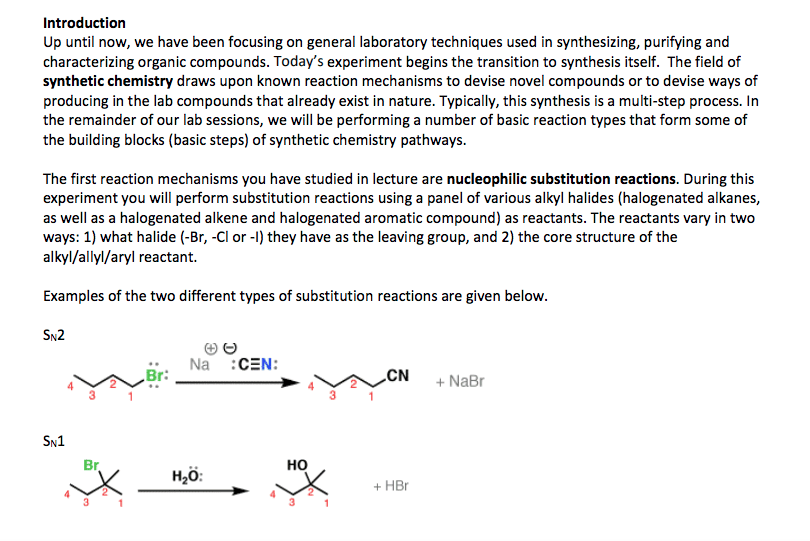
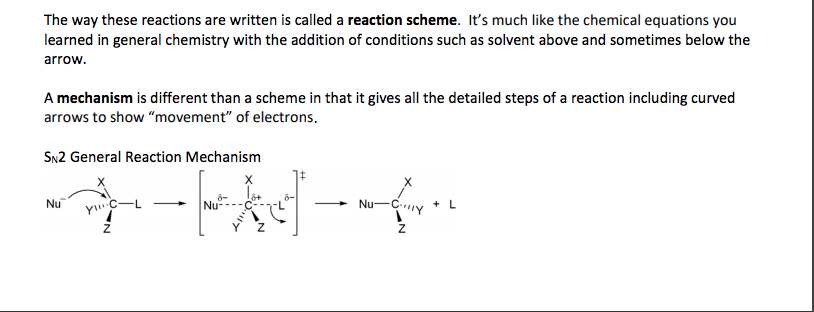
Compounds with an sp3 carbon bonded to a electronegative atom or group (e. g. as in alkyl halides) can undergo 2 types of reactions: substitution and/or elimination: The rate of a nucleophilic substitution reaction depends upon: (1) the structure of the alkyl halide (2) the leaving group (3) the nucleophile (4) the solvent. The rate of an sn2 reaction depends on the concentration of both the alkyl halide and the nucleophile, therefore it is a bimolecular reaction (second order). In an sn2 reaction the reactivity order of alkyl halides (due to steric effects) is: Sn2 reactions take place in a single step, without intermediates. The nucleophile attacks the substrate from a direction 180o away from the leaving group, causing inversion of configuration of the molecule. The leaving group in the sn2 reaction. The leaving ability of a group is a measure of its ease of displacement.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232