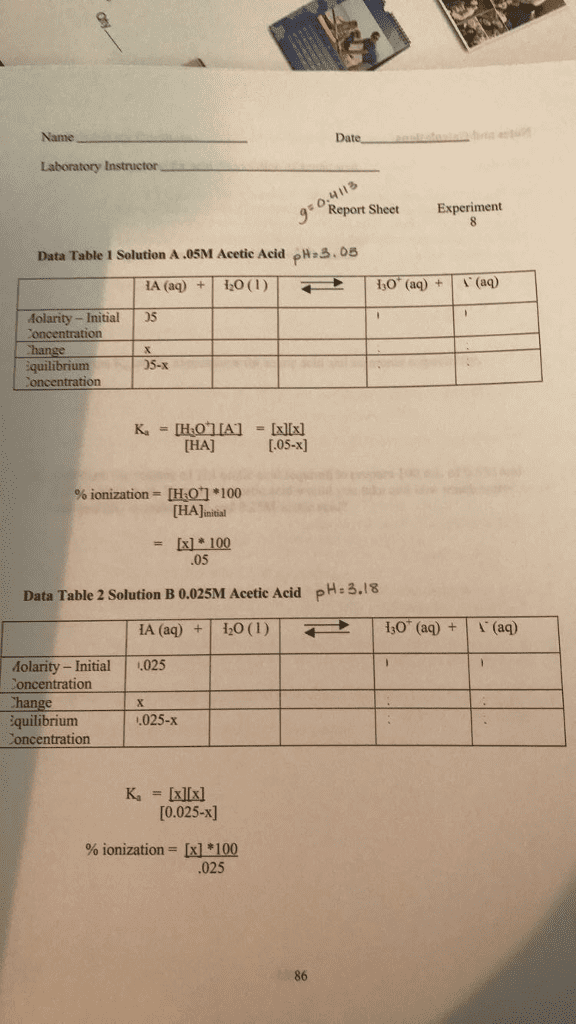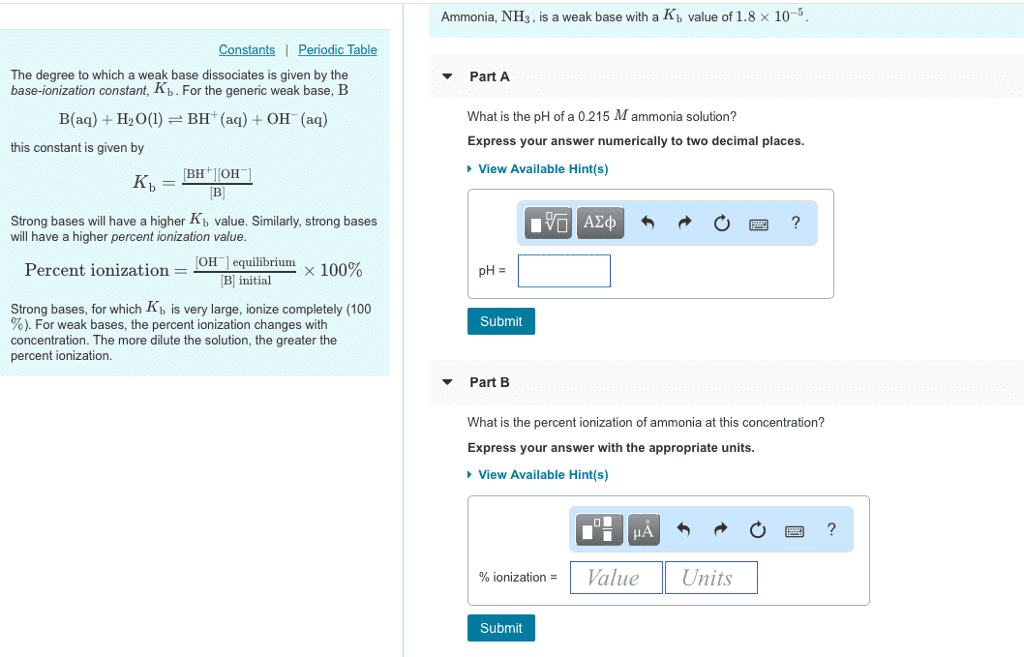
Ammonia, NH3 , is a weak base with a Kb value of 1.8 Ã 10-5 The degree to which a weak base dissociates is given by the base-ionization constant, Kb. For the generic weak base, B â¼ Part A What is the pH of a 0.215 M ammonia solution? Express your answer numerically to two decimal places. B(aq) H2(BH (aq) OH (aq) this constant is given by View Available Hint(s) Strong bases will have a higher Kb value. Similarly, strong bases will have a higher percent ioniza OH! equilibrium. Ã 100% Percent ionization B initial Strong bases, for which Kb is very large, ionize completely (100 %). For weak bases, the percent ionization changes with concentration. The more dilute the solution, the greater the percent ionization. Submit Part B What is the percent ionizatio nia at this concentration? Express your answer with the appropriate units View Available Hint(s) 1 Value Units | % ionization = Submit