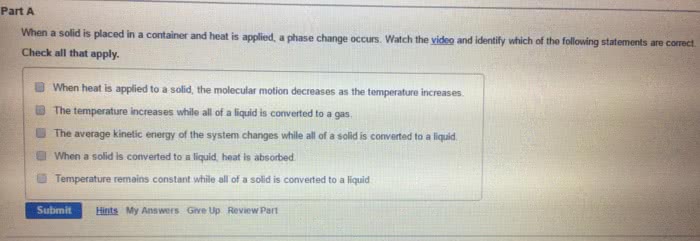
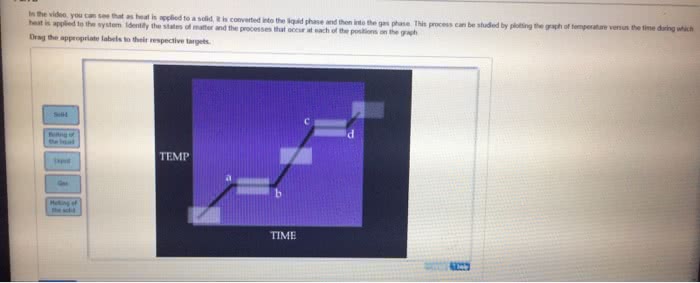
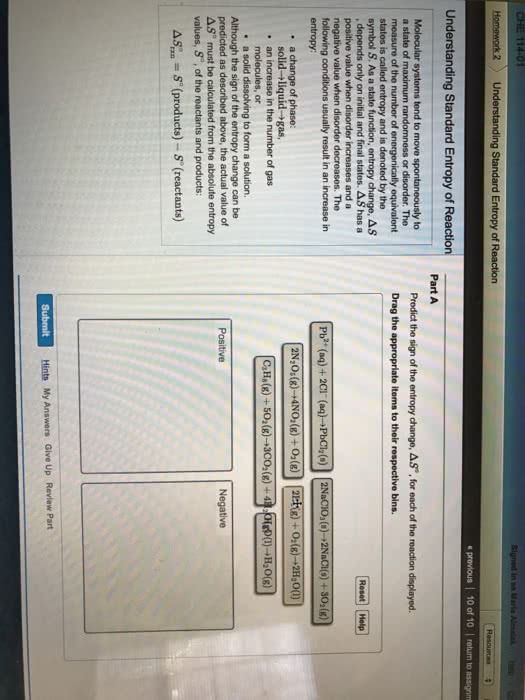
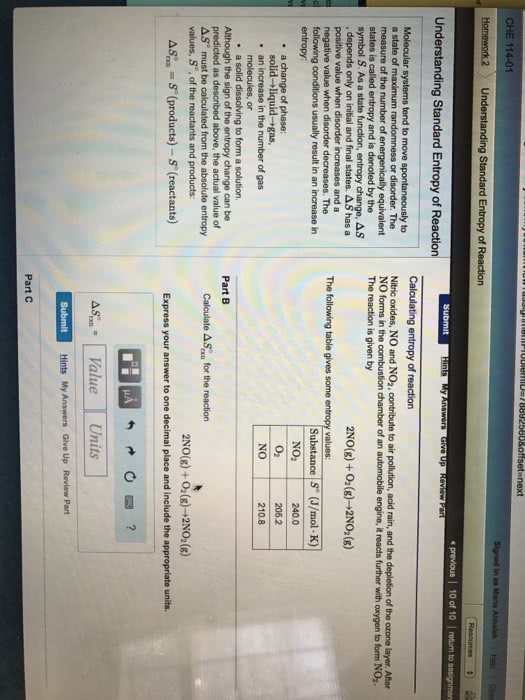
CHEM 1101 Chapter Notes - Chapter 8: Combined Gas Law, Ideal Gas Law, Kinetic Theory Of Gases
Document Summary
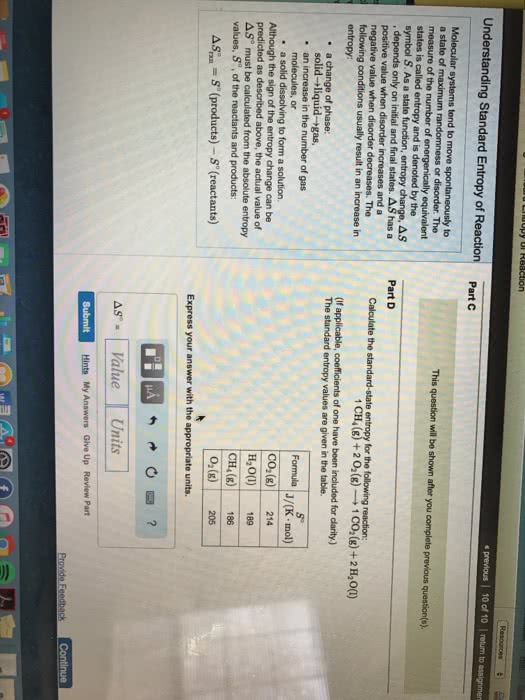
Matter exists in any of three phases, or states solid, liquid, and gas. The state depends on the relative strength of the attractive forces between particles compared to the kinetic energy of the particles. or a change of state. The transformation of a substance from one state to another is called a phase change , Every change of state is reversible and characterized by a free-energy change. H is a measure of the heat absorbed or released (enthalpy change) S is a measure of the change in molecular disorder (entropy change) The temperature at which the liquid phase is in equilibrium with the solid phase is called. The temperature at which the gas phase is in equilibrium with the liquid phase is called the melting point . the boiling point . called sublimation . Dry ice (solid co 2 ) changes directly to a gas without melting.