CHEM 0110 Chapter 7: Chpt. 7 - Quantum Theory of the Atom
Document Summary
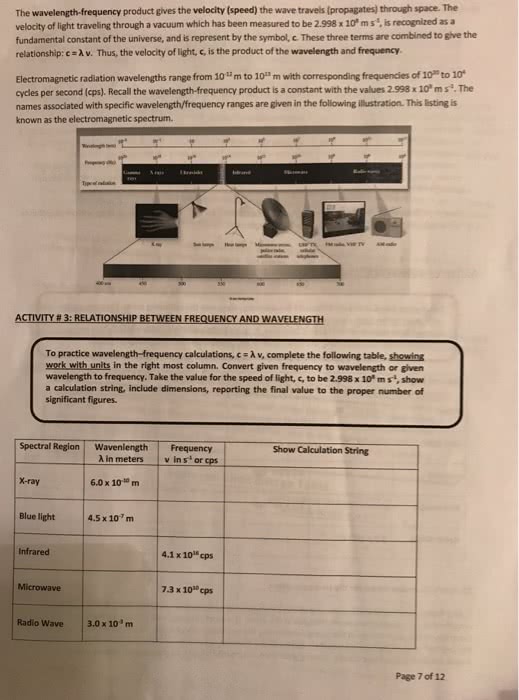
Chapter 7: quantum theory of the atom. Light waves, photons, and the bohr theory. Section 1 the wave nature of light. Wave: a continuously repeating change or oscillation in matter or in a physical field: light is a wave. Consists of oscillations in electric and magnetic fields that can travel through space. Visible light, x rays, and radio waves are all forms of electromagnetic radiation. Wavelength ( ): the distance between any two adjacent identical points of a wave: the distance between two adjacent peaks or troughs of a wave. Frequency (v): the number of wavelengths of that wave that passes a fixed point in one unit of time (usually one second: unit = /s or s-1 called hertz (hz) Electromagnetic spectrum: the range of frequencies or wavelengths of electromagnetic radiation: visible spectrum (light) Section 2 quantum effects and photons: isaac newton. Studied the properties of light in the 17th century.