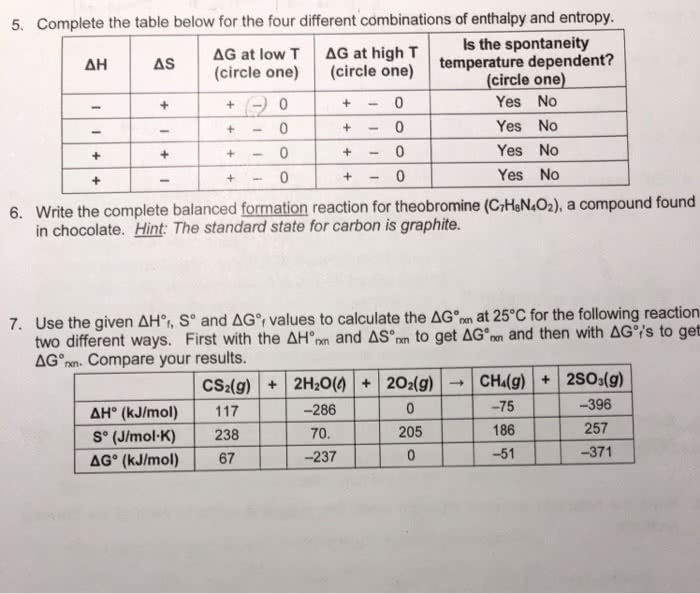
I used the equation Delta G = Delta H - T( Delta S), but I am not sure I am findind enthalpy or entropy correctly if you could please explain those steps I would apperciate it.
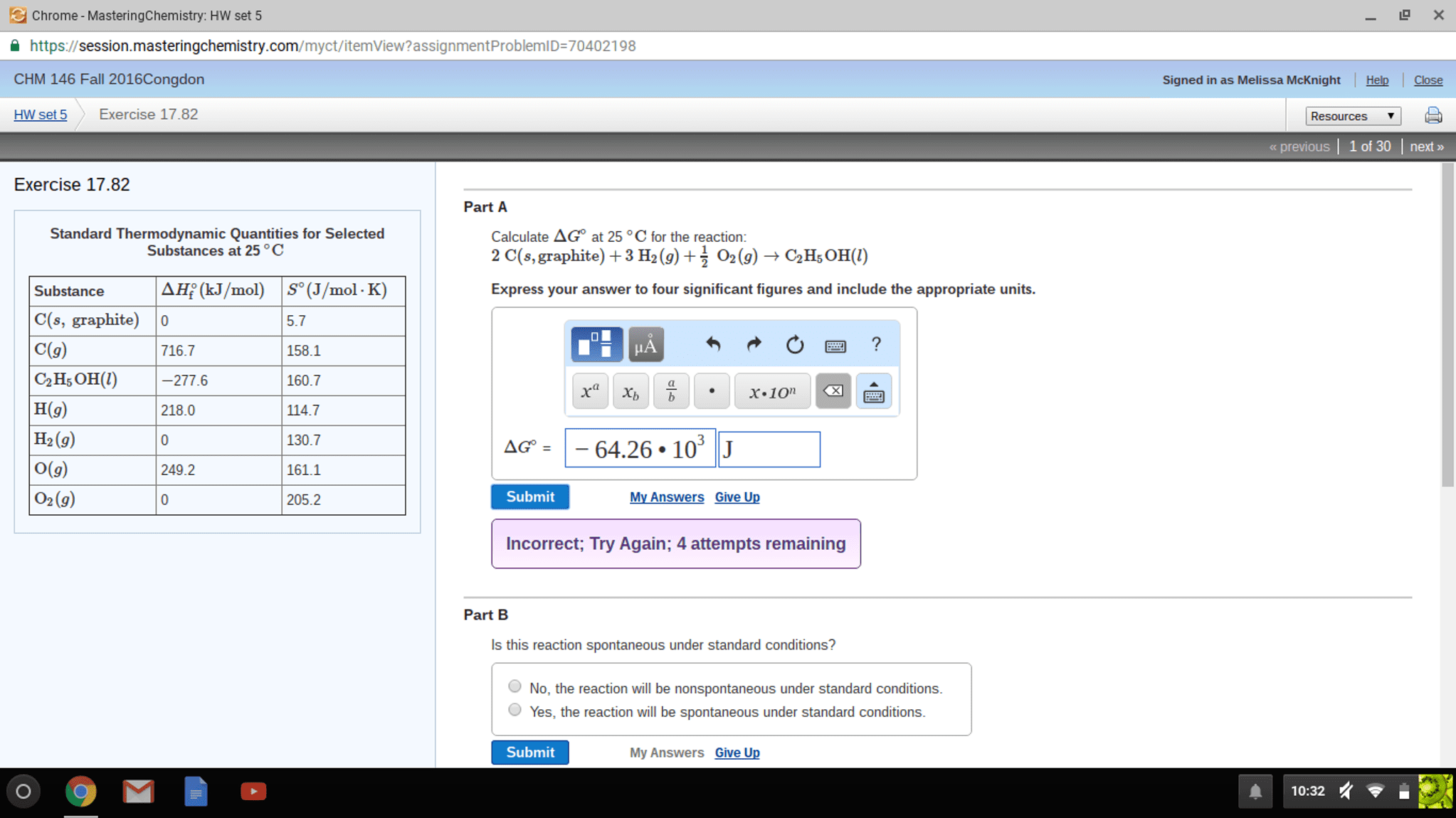
Chrome-MasteringChemistry: HW set 5 session masteringchemistry.com/ myct/itemView? assignment ProblemID 70402198 CHM 146 Fall 2016Congdon HW set 5 Exercise 17.82 Exercise 17.82 Part A Standard Thermodynamic Quantities for Selected Calculate AGO at 25 C for the reaction: Substances at 25 C 2 C (s, graphite) +3 H2 (g) O2 (g) C2H5oH() AH (kJ/mol) (J/mol. K) Express your answer to four significant figures and include the appropriate units. Substance C(s, graphite) 0 5.7 C(g) 716.7 158.1 C2H5OH(l) 277.6 160.7 H(g) 218.0 114.7 H2 (g) 130.7 AG 64.26 10 J 161.1 249.2 C2 (g) Submit My Answers Give Up 205.2 Incorrect, Try Again; 4 attempts remaining Part B ls this reaction spontaneous under standard conditions? O No, the reaction will be nonspontaneous under standard conditions Yes, the reaction will be spontaneous under standard conditions. Submit My Answers Give Up X signed in as Melissa McKnight Help l Close Resources previous l 1 of 30 l next 10:32