01:160:161 Chapter Notes - Chapter 10.3-10.4: Atmospheric Pressure, Barometer, Torr
Document Summary
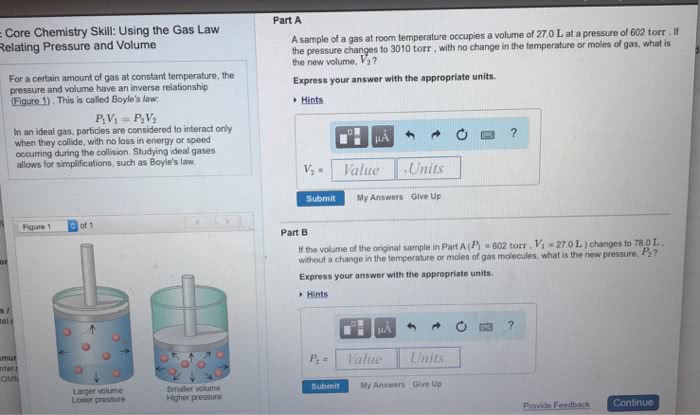
Each collision exerts only a small force, but when the forces of the many particles are summed, they quickly add up. The result of the constant collisions between the atoms or molecules in a gas and the surfaces around them is pressure. The pressure that a gas sample exerts is the force that results from the collisions of gas particles divided by the area of the surface with which they collide. The pressure exerted by a gas sample, therefore, depends on the number of gas particles in a given volume- the fewer the gas particles, the lower the force per unit area and the lower the pressure. Because the number of gas particles in a given volume generally decreases with increasing altitude, pressure decreases with increasing altitude. A common unit of pressure, the millimeter of mercury (mmhg), originates from how pressure is measured with a barometer.