CHE 106 Chapter Notes - Chapter 6: Bohr Model, Rydberg Formula, Magnetic Quantum Number

63
CHE 106 Full Course Notes
Verified Note
63 documents
Document Summary
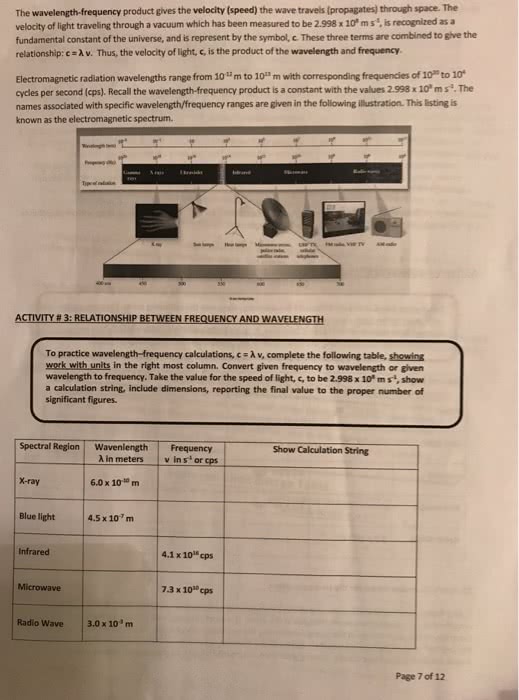
Chapter 6 reading notes: electronic structure of atoms. Electronic structure: the number of electrons in the atom as well as their distribution around the nucleus and their energies. Electromagnetic radiation: also known as radiant energy: radio waves. Infrared radiation (heat: x-rays, speed of light: 2. 998 x 10^8 m/s. Wavelength: the distance between two adjacent peaks (or troughs) All electromagnetic radiation moves at the same speed, namely, the speed of light c = Matter can emit and absorb energy only in whole number multiples of hv, 2hv, 3 hv, etc. Because the energy can be released only in specific amounts, the allowed energies are quantized their values are restricted to certain quantities. Work function: a certain amount of energy that is required for the electrons to overcome the attractive forces holding them in the metal. Depending on the situation it may behave more like one over the other. Ch 6. 3 line spectra and the bohr model.