CH 231 Chapter Notes - Chapter 1: Sigma Bond, Ionic Bonding, Lone Pair
Document Summary
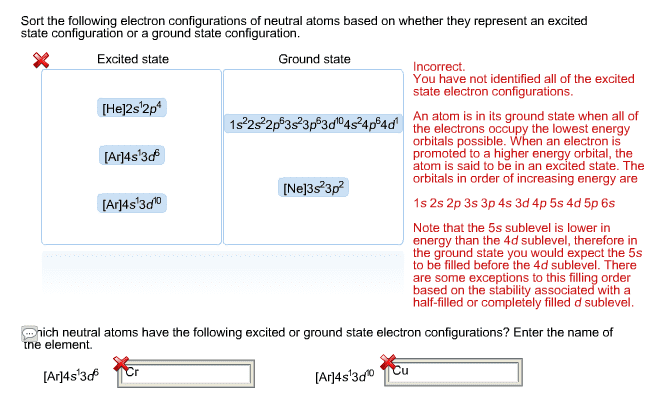
# protons # neutrons: orbitals: where the electrons are distributed outside the nucleus. P orbital: electron shells: orbitals are organized into different layers. Electron energy diagram- start at lowest energy and work up: electron configuration. 3 rules to predict the lowest energy configuration. Aufbau principle- lowest energy orbitals fill first. Pauli exclusion principle- electrons have 2 orientations ( ) and 2 electrons in an orbital must have opposite spins. Hu(cid:374)d"s rule- fill each orbital with one electron of the same spin before filling orbitals with 2 electrons. Because the resultant compound is more stable typically, energy flows out of a chemical system when bonds are formed; likewise, energy must be put into a system to break bonds: the octet rule. 8 electrons in the valence shell = stability: ionic bonding. Transferring of electrons leads to attractions between cations and anions: covalent bond. Sharing of electrons between atoms: lewis structure. Valence electrons are shown as dots: kekul structures.