CHEM 1062 Chapter Notes - Chapter 18: Conjugate Acid, Acid Strength, Ph Indicator
Document Summary
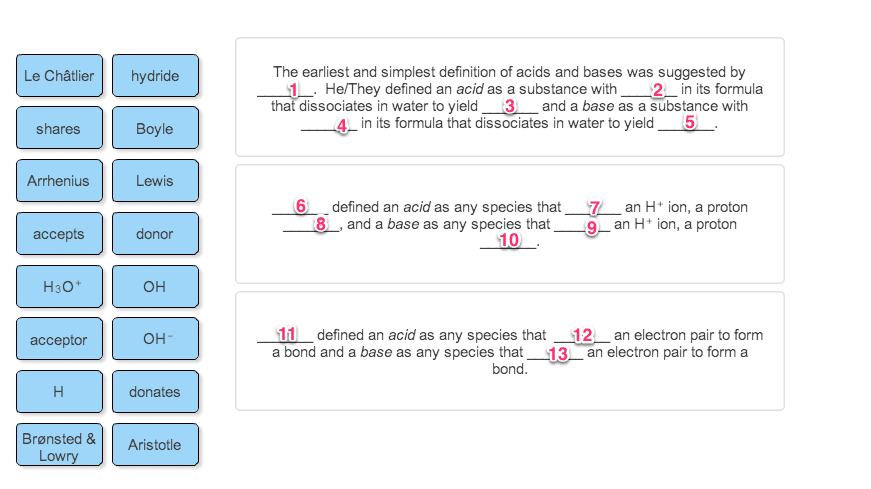
Water is the product in all reaction between strong acids and strong bases. When an acid dissociates in water, solvent molecules participate in the reaction: Ha + h2o --- > a- + h3o+; water molecules surround the proton to form h bonded species. The proton"s charge density is so high that it attracts water and covalently bonds to one of the lone electron pairs of a water molecule"s o making hydronium. Earliest acid/base definition was the arrhenius definition which classifies in terms of formulas and behaviour in water. An acid is a substance with h in its formula that dissociates in water to yield h3o+ A base is a substance with oh in its formula that dissociates in water to yield oh- Arrhenius acids never contain discrete h+ ions; instead they have covalently bonded h atoms that ionize when the molecules dissolves in water.