CHEM 103 Chapter Notes - Chapter M7Q6: George Uhlenbeck, Pauli Exclusion Principle, Samuel Goudsmit
Document Summary
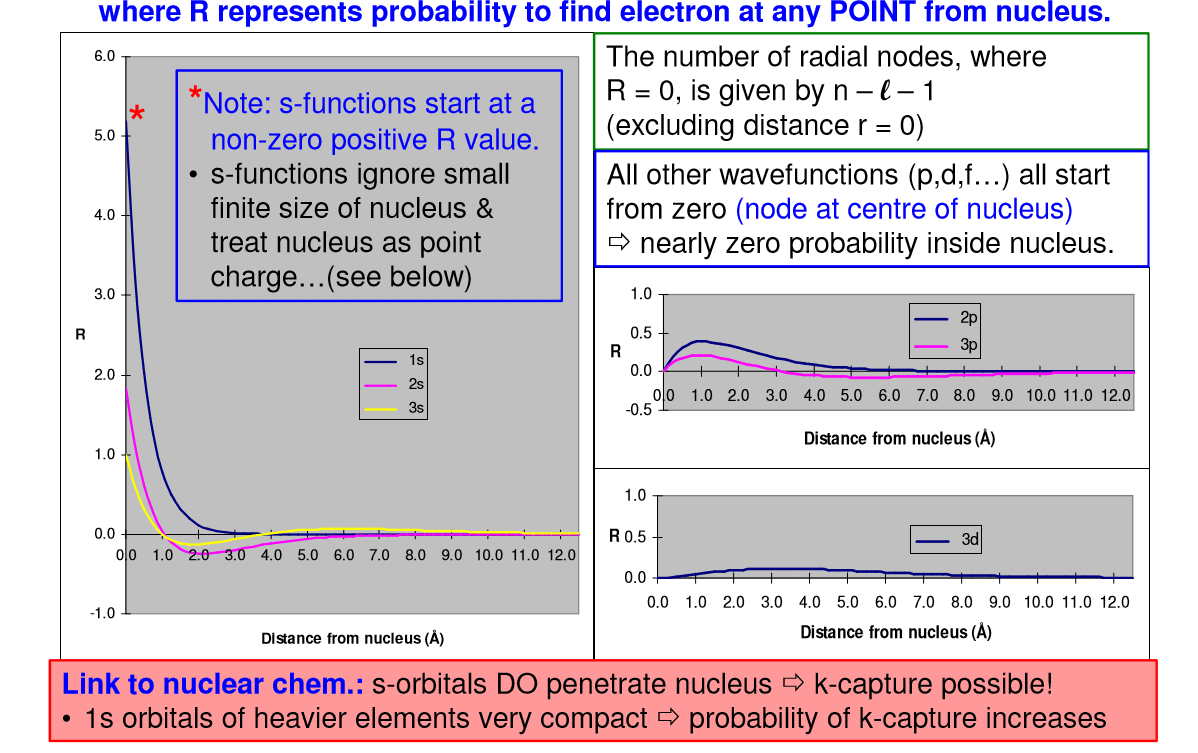
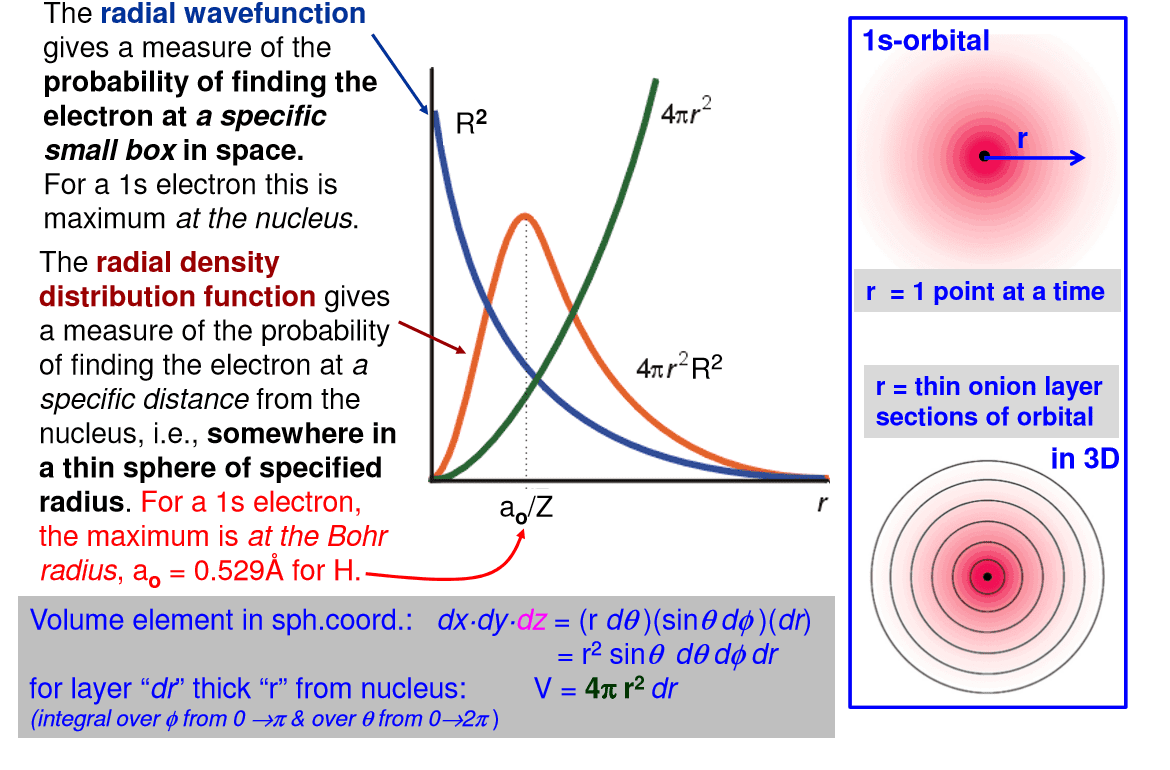
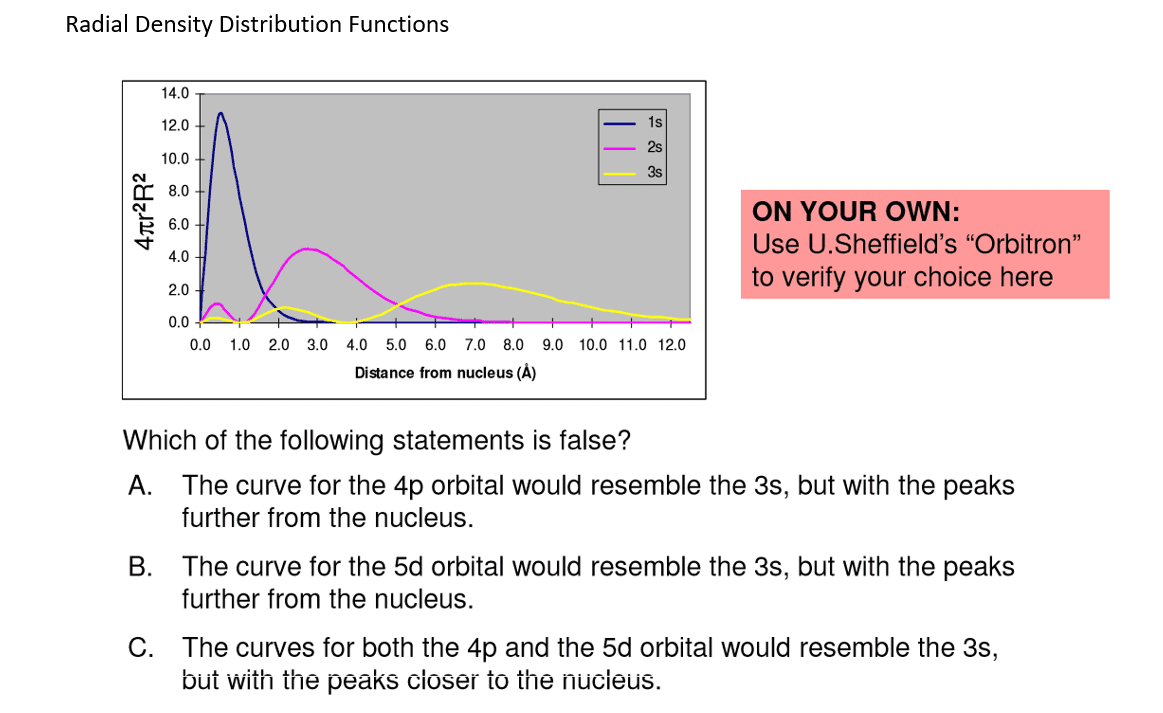
4th quantum number (m7q6: spherical surface area, surface area of each spherical shell = 4 r2. Three things happen to s orbitals as n increases : they become larger, extending farther from the nucleus, they contain more nodes. This is similar to a standing wave that has regions of significant amplitude separated by nodes (points with zero amplitude). The number of radial nodes in an orbital is n l 1: for a given atom, the s orbitals also become higher in energy as n increases because of the increased distance from the nucleus. Iv. p-orbitals: only s orbitals are spherically symmetrical. As the value of l increases, the number of orbitals in a given subshell increases, and the shapes of the orbitals become more complex. 1, with three values of ml ( 1, 0, and +1), there are three 2p orbitals: the size and complexity of the p orbitals for any atom increase as the principal quantum number n increases.