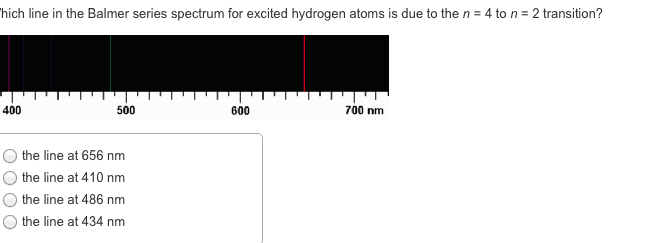
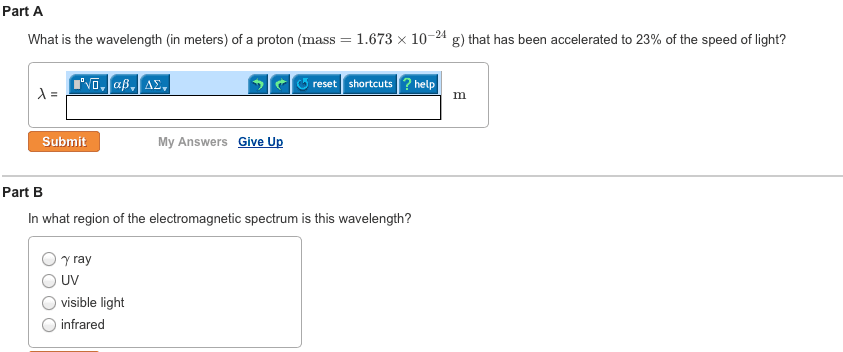
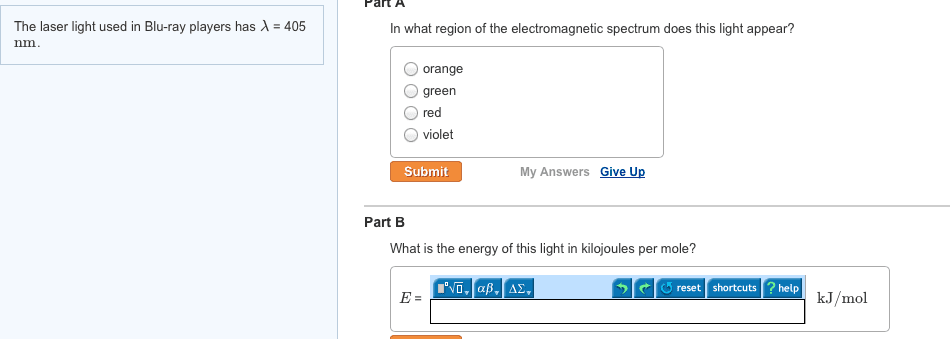
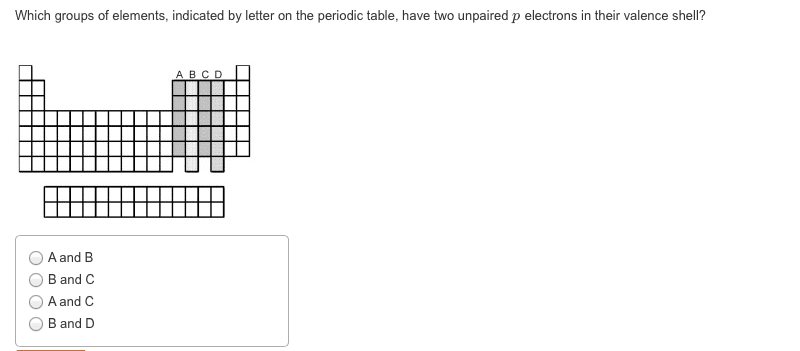CHEM 1A03 Lecture 3: Unit 3 Module 1: Atomic Structure and Theory

35
CHEM 1A03 Full Course Notes
Verified Note
35 documents
Document Summary
Unit 3 module 1: atomic structure and theory. Identify the relationships between wavelength, frequency, and energy for light. Discuss the wave-particle duality of light and of the electron, and describe the experimental evidence and theoretical basis for their duality. Qualitatively and quantitatively describe energy transitions (absorption and emission) in the hydrogen atom. Use the four quantum numbers to describe atomic orbitals and electrons in orbitals and write electron configurations. All light is made up of electromagnetic waves. The wave is composed of an electrical component in one direction, and a magnetic component perpendicular to it, also travelling in the same direction. The distance between the peaks and troughs is the wavelength, . How many peaks go by in a given amount of time is frequency, v. We get the relationship between frequency and wavelength, where c = v. C is a constant, it is the speed of light.